
A Household Ingredient May Revolutionize Polyester Recycling, Making It Simpler and Greener
Polyester ranks as the world’s second most used textile, however, it is an environmental menace, especially because most of it never gets recycled. The fabric, a blend of plastic and cotton, has been difficult for the industry to separate and therefore recycle. However, a group of young chemists from the University of Copenhagen has invented a green and surprisingly simple solution using a single household ingredient.
From clothes to sofas to curtains, polyester dominates our everyday lives, with a staggering 60 million tons of this popular fabric produced annually. However, polyester production takes a toll on the climate and the environment, as only a mere 15% of it is recycled, while the rest ends up in landfills or incinerated, being responsible for more carbon emissions.
Recycling polyester poses a significant challenge, particularly in separating the plastic and cotton fibers that the blend fabric is made of without losing either of them in the process. Conventional recycling methods often prioritize preserving the plastic component, resulting in a loss of cotton fibers. Moreover, these methods are costly, complex, and generate metal waste due to the use of metal catalysts, which can be cytotoxic and contaminate the process.

In a remarkable breakthrough, a group of young chemists has unveiled a surprisingly simple solution to this pressing problem, potentially revolutionizing the sustainability of the textile industry.
“The textile industry urgently requires a better solution to handle blended fabrics like polyester/cotton. Currently, there are very few practical methods capable of recycling both cotton and plastic—it’s typically an either-or scenario. However, with our newly discovered technique, we can depolymerize polyester into its monomers while simultaneously recovering cotton on a scale of hundreds of grams, using an incredibly straightforward and environmentally friendly approach. This traceless catalytic methodology could be the game-changer,” explains postdoc Yang Yang of the Jiwoong Lee group at the University of Copenhagen’s Department of Chemistry, who serves as the lead author of the scientific research article.
Hartshorn Salt and 24 Hours in the ‘Oven’
The new method requires no special equipment – just heat, a non-toxic solvent, and an ordinary household ingredient.
“For example, we can take a polyester dress, cut it up into small pieces, and place it in a container. Then, add a bit of mild solvent, and thereafter hartshorn salt, which many people know as a leavening agent in baked goods. We then heat it all up to 160 degrees Celsius and leave it for 24 hours. The result is a liquid in which the plastic and cotton fibers settle into distinct layers. It’s a simple and cost-effective process,” explains Shriaya Sharma, a doctoral student of the Jiwoong Lee group at the Department of Chemistry and study co-author.
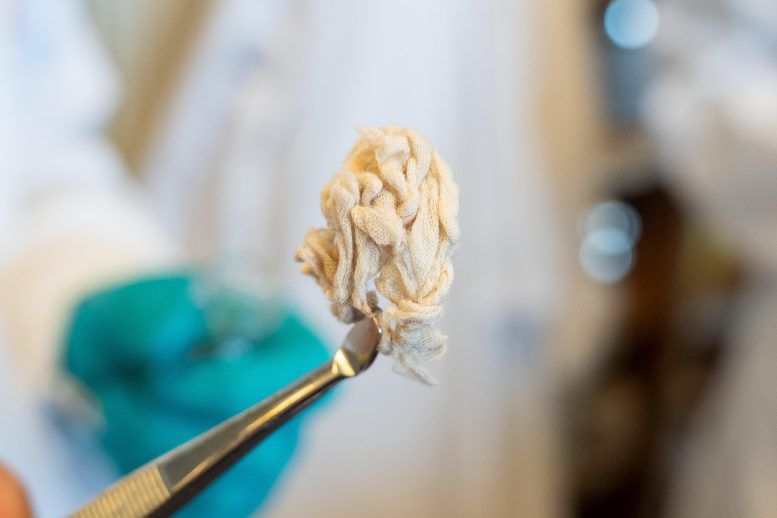
In the process, the hartshorn salt, also called ammonium bicarbonate, is broken down into ammonia, CO2, and water. The combination of ammonia and CO2 acts as a catalyst, triggering a selective depolymerization reaction that breaks down the polyester while preserving the cotton fibers. Although ammonia is toxic in isolation, when combined with CO2, it becomes both environmentally friendly and safe for use. Due to the mild nature of the chemicals involved, the cotton fibers remain intact and in excellent condition.
Previously, the same research group demonstrated that CO2 could serve as a catalyst for breaking down nylon, among other things, without leaving any trace. This discovery inspired them to explore the use of hartshorn salt. Nevertheless, the researchers were pleasantly surprised when their simple recipe yielded successful results.
“At first, we were excited to see it work so well on the PET bottles alone. Then, when we discovered that it worked on polyester fabric as well, we were just ecstatic. It was indescribable. That it was so simple to perform was nearly too good to be true,” says Carlo Di Bernardo, doctoral student and study co-author.

While the method has only been tested at the laboratory level thus far, the researchers point to its scalability and are now in contact with companies to test the method on an industrial scale.
“We’re hoping to commercialize this technology that harbors such great potential. Keeping this knowledge behind the walls of the university would be a huge waste,” concludes Yang Yang.

Pet and Polyester
PET is the most widely used type of plastic in the world. The production of PET, a plastic most familiar to us as beverage bottles, amounts to 70 million tons annually and is growing every year. One-third of global PET production is used to produce polyester and other synthetic textiles.
Also Works on Waste
The new recycling method based on hartshorn salt (ammonium bicarbonate) works on PET plastic alone, as well as on PET and cotton blended materials.
“If we throw dirty plastic waste in a container, we still get good quality cotton and plastic monomer out of it. This can even be a plastic bottle with juice residue still in it. We just put it in and begin the reaction. It still works,” says Shriaya Sharma.
Reference: “Catalytic Fabric Recycling: Glycolysis of Blended PET with Carbon Dioxide and Ammonia” by Yang Yang, Shriaya Sharma, Carlo Di Bernardo, Elisa Rossi, Rodrigo Lima, Fadhil S. Kamounah, Margarita Poderyte, Kasper Enemark-Rasmussen, Gianluca Ciancaleoni and Ji-Woong Lee, 17 July 2023, ACS Sustainable Chemistry & Engineering.
DOI: 10.1021/acssuschemeng.3c03114
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Why does this scientific article continually refer to “hartshorn salt” instead of using the compound’s correct name – ammonium carbonate, which is only mentioned once???
I have a BSc and a PhD in chemistry, specialising in inorganic chemistry and have been a university and college lecturer. Yet even I had to google the term hartshorn salt to find out what on earth the article was talking about! Okay, half way through it does mention the correct name. But come on, a science article using a weird obsolete ancient historic name instead of just ammonium carbonate does not make sense.