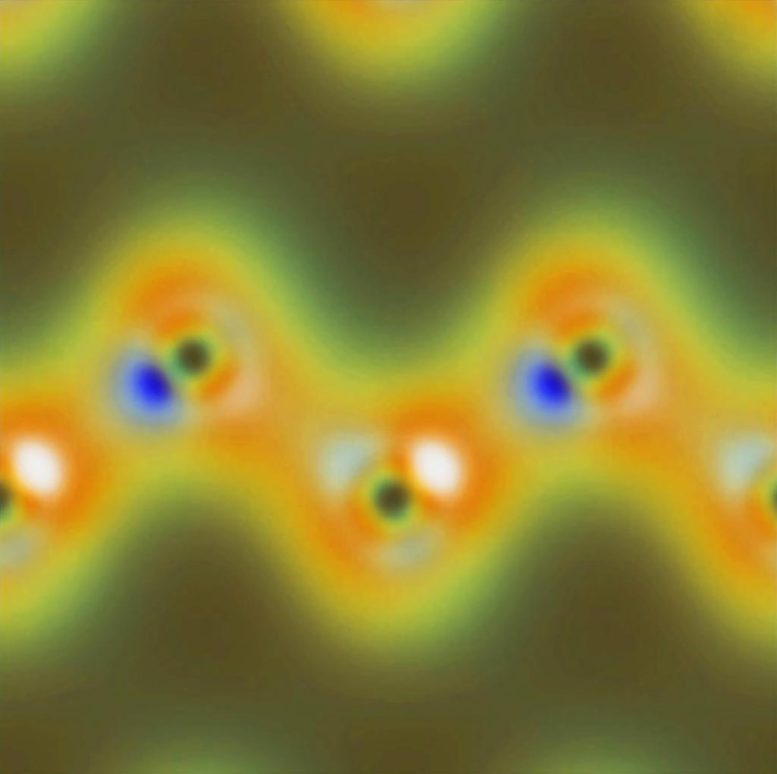
By mixing a pulse of X-rays with a pulse of lower-frequency optical light waves, a team of scientists at the Berkley Lab was able to measure the optical manipulation of chemical bonds at the atomic scale.
Light changes matter in ways that shape our world. Photons trigger changes in proteins in the eye to enable vision; sunlight splits water into hydrogen and oxygen and creates chemicals through photosynthesis; light causes electrons to flow in the semiconductors that make up solar cells; and new devices for consumers, industry, and medicine operate with photons instead of electrons. But directly measuring how light manipulates matter on the atomic scale has never been possible, until now.
An international team of scientists led by Thornton Glover of the U.S. Department of Energy’s Lawrence Berkeley National Laboratory (Berkeley Lab) used the Linac Coherent Light Source (LCLS) at the SLAC National Accelerator Laboratory to mix a pulse of superbright X-rays with a pulse of lower frequency, “optical” light from an ordinary laser. By aiming the combined pulses at a diamond sample, the team was able to measure the optical manipulation of chemical bonds in the crystal directly, on the scale of individual atoms.
The researchers report their work in the August 30, 2012 issue of the journal Nature.
Mixing X-rays with light in X-ray diffraction
X-ray and optical wave-mixing is an X-ray diffraction technique similar to that long used in solving the structures of proteins and other biological molecules in crystalline form. But in contrast to conventional diffraction, wave mixing selectively probes how light reshapes the distribution of charge in a material. It does this by imposing a distinction between X-rays scattered from optically perturbed charge and X-rays scattered from unperturbed charge.
“You can think of the electrons orbiting atoms in a material as belonging to one of two groups,” says Glover. “The ‘active’ electrons are the outer, loosely bound valence electrons that participate in chemical reactions and form chemical bonds. The ‘spectator’ electrons are the ones tightly wrapped around the nucleus at the atom’s core.”
Glover explains that “because the x-ray photon energy is large compared to the electron binding energy, in a typical scattering experiment all electrons scatter with comparable strength and are therefore more or less indistinguishable.” The core-electron signal usually swamps the weaker valence-charge signal because there are many more core electrons than valence electrons.
“So x-rays can tell you where atoms are, but they usually can’t reveal how the chemically important valence charge is distributed,” Glover says. “However, when light is also present with the X-rays, it wiggles some portion of the chemically relevant valence charge. X-rays scatter from this optically driven charge, and in doing so the X-ray photon energy is changed.”
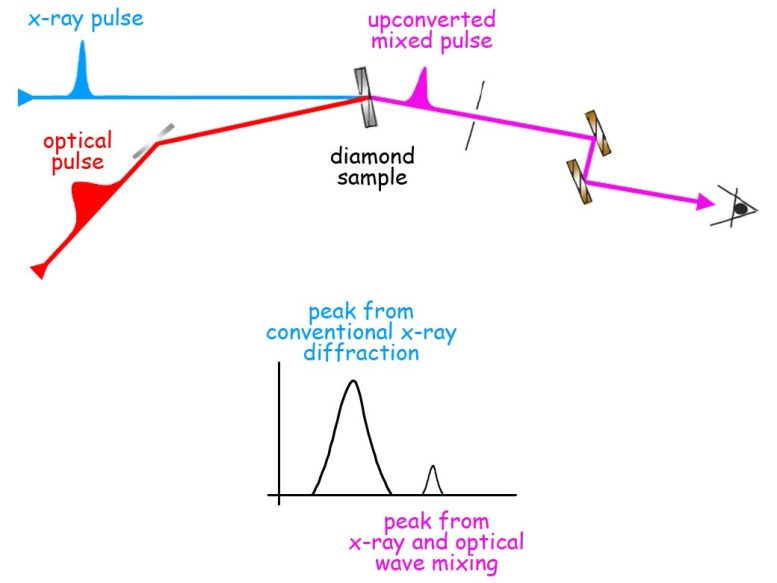
The modified X-rays have a frequency (or energy) equal to the sum of the frequencies of both the original X-ray pulse and the overlapping optical pulse. The change to a slightly higher energy provides a distinct signature, which distinguishes wave-mixing from conventional X-ray diffraction.
“Conventional diffraction does not provide direct information on how the valence electrons respond to light, nor on the electric fields that arise in a material because of this response,” says Glover. “But with x-ray and optical wave mixing, the energy-modified x-rays selectively probe a material’s optically responsive valence charge.”
Beyond the ability to directly probe atomic-scale details of how light initiates such changes as chemical reactions or phase transitions, sensitivity to valence charge creates new opportunities to track the evolution of chemical bonds or conduction electrons in a material – something traditional x-ray diffraction does poorly. Different components of the valence charge can be probed by tuning the so-called optical pulse; higher-frequency pulses of extreme ultraviolet light, for example, probe a larger portion of valence charge.
Because mixing X-ray and optical light waves creates a new beam, which shows up as a slightly higher-energy peak on a graph of X-ray diffraction, the process is called “sum frequency generation.” It was proposed almost half a century ago by Isaac Freund and Barry Levine of Bell Labs as a technique for probing the microscopic details of light’s interactions with matter, by separating information about the position of atoms from the response of valence charge exposed to light.
But sum frequency generation requires intense X-ray sources unavailable until recently. SLAC’s LCLS is just such a source. It’s a free-electron laser (FEL) that can produce ultrashort pulses of high-energy “hard” x-rays millions of times brighter than synchrotron light sources, a hundred times a second.
“The breadth of the science impact of LCLS is still before us,” says Jerome Hastings, a professor of photon science at the LCLS and an author of the Nature article. “What is clear is that it has the potential to extend nonlinear optics into the x-ray range as a useful tool. Wave mixing is an obvious choice, and this first experiment opens the door.”
Diamonds are just the beginning
Glover’s team chose diamond to demonstrate x-ray and optical wave mixing because diamond’s structure and electronic properties are already well known. With this test bed, wave mixing has proved its ability to study light-matter interactions on the atomic scale and has opened new opportunities for research.
“The easiest kinds of diffraction experiments are with crystals, and there’s lots to learn,” Glover says. “For example, light can be used to alter the magnetic order in advanced materials, yet it’s often unclear just what the light does, on the microscopic scale, to initiate these changes.”
Looking farther ahead, Glover imagines experiments that observe the dynamic evolution of a complex system as it evolves from the moment of initial excitation by light. Photosynthesis is a prime example, in which the energy of sunlight is transferred through a network of light-harvesting proteins into chemical reaction centers with almost no loss.
“Berkeley Lab’s Graham Fleming has shown that this virtually instantaneous energy transfer is intrinsically quantum mechanical,” Glover says. “Quantum entanglement plays an important role, as an excited electron simultaneously samples many spatially separated sites, probing to find the most efficient energy-transfer pathway. It would be great if we could use x-ray and optical wave mixing to make real-space images of this process as it’s happening, to learn more about the quantum aspects of the energy transfer.”
Such experiments will require high pulse-repetition rates that free electron lasers have not yet achieved. Synchrotron light sources like Berkeley Lab’s Advanced Light Source, although not as bright as FELs, have inherently high repetition rates and, says Glover, “may play a role in helping us assess the technical adjustments needed for high repetition-rate experiments.”
Light sources with repetition rates up to a million pulses per second may someday be able to do the job. Glover says, “FELs of the future will combine high-peak brightness with high repetition rate, and this combination will open new opportunities for examining the interactions of light and matter on the atomic scale.”
Reference: “X-ray and optical wave mixing” by T. E. Glover, D. M. Fritz, M. Cammarata, T. K. Allison, Sinisa Coh, J. M. Feldkamp, H. Lemke, D. Zhu, Y. Feng, R. N. Coffee, M. Fuchs, S. Ghimire, J. Chen, S. Shwartz, D. A. Reis, S. E. Harris and J. B. Hastings, 29 August 2012, Nature.
DOI: 10.1038/nature11340
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Citation: Design and development of multi optic wave pi phase selective modulator in genetic corrections for new Chemo therapy machine
High pulse-repetition rates that free electron lasers have that may be achieved by using x ray-photon triggered dynamics that may be achieved for a new method of laser beam in understanding dual forces operative in super nova new type of genetic correction waves may be generated
Light changes matter in ways that shape our world. Photons trigger changes in proteins in the eye to enable vision; sunlight splits water into hydrogen and oxygen and creates chemicals through photosynthesis; light causes electrons to flow in the semiconductors that make up solar cells; and new devices for consumers, industry, and medicine operate with photons instead of electrons. But directly measuring how light manipulates matter on the atomic scale has never been possible, until now.
Linus Pauling Professor of Chemical Engineering, Bioengineering, and Biochemistry at the California Institute of Technology along with Sankaravelayudhan Nandakumar of Hubble research fellow formerly with Anna University Chennai Assistant professor and Project coordinator in windmill technology retired as chief Engineer Thermal power Station former Alumini of College of engineering Trivandrum A CO Associate coroordinator with Hon.Stephaen hawkings of Applied mathematics dept Cambridge University
Citation:The cross dynamical pi phase opertaive between large module converged aas s mall in between the photon pi dynmaics oprtaive between magnetic and electricfield as applicable in ultraviolet rays breaking the geneticgain suddenly by analogy of electron spin axis pi shifted wit reference to pole axis an oscilation that brings out sudden changs perhaps applicable in gravit waves as deviated from Lobochensky –Graffe dynamics as the poit vector traces bywith refrence to Uranus spin axis pi shifted with refrence to pole axis as atwin brother action of saturn requiring more information and collections.This may aslo be operative in understanding while the ultraviolet rays that initiates such a spin as root locus trnsfer factor polynominial dynmaics paving the way for Gaussian field chnages.
Ellipsoidal expansion may be contributed on ATGU bases spirality followed by an ejection and chemical bonding contributes to genetic continuity
In physics, the Poynting vector represents the directional energy flux (the energy transfer per unit area per unit time) of an electromagnetic field. The SI unit of the Poynting vector is the watt per square metre (W/m2). It is named after its discoverer John Henry Poynting who first derived it in 1884.[1]:132 Oliver Heaviside also discovered it independently. We would thus not be considering the instantaneous E(t) and H(t) used above, but rather a complex (vector) amplitude for each which describes a coherent wave’s phase (as well as amplitude) using phasor notation. Note that these complex amplitude vectors are not functions of time, as they are understood to refer to oscillations over all time. A phasor such as is understood to signify a sinusoidally varying field whose instantaneous amplitude E(t) follows the real part of Em e jwt and w is the (radian) frequency of the sinusoidal wave being considered which seems to follow a resonance and anti resonance . Poynting vector (S, blue) in the space surrounding it, along with the fields it is derived from; the electric field (E, red) and the magnetic field (H, green). In the region around the battery the Poynting vector is directed outward, indicating power flowing out as the refractive index by permeability change between positive and negative. An information available from Euler formula may be applied in complex frequency that act along polynomial forming an imaginary pole along JW axis a theoretical system that may be operated as afeedback frequency selective Nyquist oscillation as it passes through unit circle at 1,j0that follw a pole and xero oscillation that becomes regerative.While s is analagoud to frequency and s is a coplex sytem that follow a dampnss function as rho+jw as w alone id the positive real frequency a generating concept that follow e st that must have apaired conjugate as complex poles represent a forming complex constant amplitude sinusoidal response and appear on the j axis also.Hence they follw an Ruler formula by analogy in understanding Laplace transform theory forming a F(w) scalar factor that swing between poles and zeros ,this means it becomes conductive or non=conductive as the case may be sometimes becomes supe conductive based on the pointing vector that swing along magnetic electric synchronise to become frequenct selective spiraality comparable with that of a galaxy expansion
The electricfield becomes a pole and zero by alternative frequency selective configuration.
An interesting polar nucleotides producing selective pointing vector along the plane of variable frequency tilting angular selection that vary between phase velocity becoming anti parallel and parallel vary the frequency selective antenna forming an enlarging polar nucleotides spirally having a pointing vector selective mutation. Crick’s ATGU bases in polar nucleotides may follow a ellipsoidal spirality expansion by an ejection forming a continuous mutations as observed in flower petals that really follow a selective frequency of rotation in frequency phases follow a pi dynamics in between zero to 360 degree rotations by a selection of frequency selective by a pointing vector operative continue the journey of genetic continuity a very important deviation from Crick’s observation requiring the various mutation of continuity says Sankaravelayudhan Nandakumar. What is inside galaxy twisting has got a polar nucleotides twists by an analogy. This means electrons circulating around a nucleus us does not follow an exact elliptical circulation may follow an expanding ellipse that change to follow a galaxy expansion by analogy.
The importance of pointing vector separating a dipole domain into a xero pole domain of dual oscillation is an important discoveryThe conclusion of the papa is there seems to be a pointing vector in between electric and magnetic field that becomes a frequency selective antenna . near the surface of of polar nucleotides Poynting vector enters direction that is almost perpendicular to the surface and swing for parallel selective as the frequency of oscillation vary between resonance and antiresonance. A pointing vector when becomes antiparallel with phase velocity it becomes invisible but when parallel becomes a visibility a possible important conclusion.
A pointing vector when becomes antiparallel with phase velocity it becomes invisible but when parallel becomes a visibility a possible production of waves more than the speed of Light may be a Tachyon at criticality. As the antenna may be partially reflective by a tilting angle the antenna becomes a degree selective during a rotational telescope. Perhaps this may contribute for unit circle interacting along frequency at 180 degrees a control of gain over amplification over frequency ranges as transformer resonance a condition for stability without regeneration in between middle frequency the forward and backward oscillation may cause a negative refractive index and positive refractive index and it becomes a degenerative forming scale factor multiplier also.It becomes a matter wave to pilot or guide that may seed the theory of wormhole
Ref:[email protected]
Citation:A pi phased genetic observation varied along the intensity of brightness betweeen Jupiter emission acting as micro lensing pointing in between the 5t plane 7th plane and 9th phase plane pointing vector vary the brightness of sun as it moves through various quantum sector