UC San Diego researchers have resolved a decades-long debate about the role of the first Crohn’s disease gene linked to an increased risk of developing the autoimmune condition.
The human digestive system relies on two main kinds of macrophages, a type of specialized white blood cell that helps keep the gut healthy. One group, known as inflammatory macrophages, protects the body by fighting off harmful microbes.
The other group, called non-inflammatory macrophages, helps heal and restore damaged tissues. When this balance is disrupted, as in Crohn’s disease (a form of inflammatory bowel disease, or IBD), the result can be persistent inflammation in the intestines, leading to pain, tissue damage, and other symptoms.
Scientists at the University of California San Diego School of Medicine have created an innovative method that combines artificial intelligence (AI) with advanced molecular biology to uncover what causes a macrophage to act as either a fighter or a healer.
Their research also provides new insight into a long-standing question about a gene known as NOD2. Discovered in 2001, NOD2 was the first gene identified as increasing the risk of developing Crohn’s disease, and it plays a key role in determining how immune cells behave in the gut.
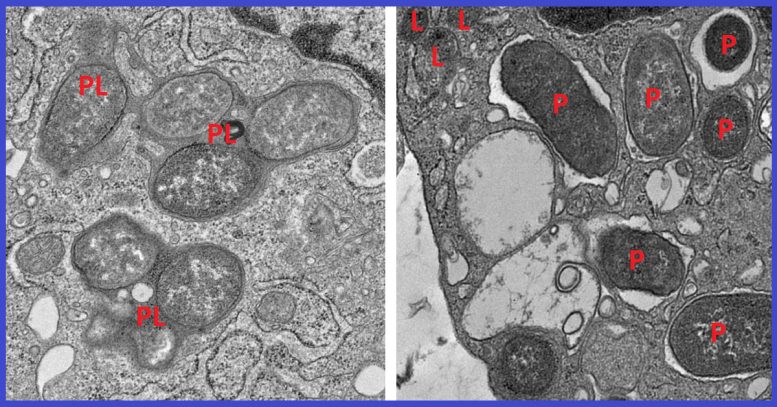
A Gene Signature That Distinguishes Macrophage Types
Using a powerful machine learning tool, the researchers analyzed thousands of macrophage gene expression patterns from colon tissue affected by IBD and from healthy colon tissue. They identified a macrophage gene signature consisting of 53 genes that reliably separates reactive, inflammatory macrophages from tissue-healing macrophages.
One of these 53 genes encodes a protein called girdin. Further analysis revealed that in non-inflammatory macrophages, a specific region of the NOD2 protein binds to girdin. This suppresses runaway inflammation, clears harmful microbes, and allows for the repair of tissues damaged by IBD. But the most common Crohn’s disease mutation to the NOD2 gene deletes the section of the gene that girdin would normally bind to. This results in a dangerous imbalance between inflammatory and non-inflammatory macrophages.

“NOD2 functions as the body’s infection surveillance system,” said senior author Pradipta Ghosh, M.D., professor and cellular and molecular medicine at UC San Diego School of Medicine. “When bound to girdin, it detects invading pathogens and maintains gut immune balance by swiftly neutralizing them. Without this partnership, the NOD2 surveillance system collapses.”
Confirming the NOD2–Girdin Link in Mice
The researchers then confirmed the importance of the interaction between NOD2 and girdin by comparing mouse models of Crohn’s disease lacking the girdin protein to those with girdin intact. They found that mice without girdin suffered an imbalance in their gut microbiome and developed inflammation of the small intestine. They often died of sepsis, a condition in which the immune system mounts an excessive response to an infection, causing inflammation throughout the body and damage to vital organs.
The gut is a battlefield, and macrophages are the peacekeepers,” said co-first author Gajanan D. Katkar, Ph.D., assistant project scientist at UC San Diego School of Medicine. “For the first time, AI has allowed us to clearly define and track the players on two opposing teams.”
By uniting AI-driven classification, mechanistic biochemistry, and mouse models, the study resolves one of the longest-running debates in Crohn’s disease. The findings not only explain how a key genetic mutation drives the disease but could also contribute to the development of treatments aimed at restoring the relationship between girdin and NOD2.
Reference: “Distinct colitis-associated macrophages drive NOD2-dependent bacterial sensing and gut homeostasis” by Gajanan D. Katkar, Mahitha Shree Anandachar, Stella-Rita C. Ibeawuchi, Ella G. McLaren, Megan L. Estanol, Kennith Carpio-Perkins, Shu-Ting Hsu, Celia R. Espinoza, Jane E. Coates, Yashaswat S. Malhotra, Madhubanti Mullick, Vanessa Castillo, Daniella Vo, Saptarshi Sinha and Pradipta Ghosh, 2 October 2025, The Journal of Clinical Investigation.
DOI: 10.1172/JCI190851
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.