
A long-standing challenge in cancer drug manufacturing may be shifting.
An international team of scientists has reported a major advance in how doxorubicin, a widely used chemotherapy drug, can be produced. Their work pinpoints and overcomes key molecular “bottlenecks” that have restricted natural production of the drug for more than 50 years.
First approved in the 1970s, doxorubicin remains a central treatment for cancers such as breast and bladder cancer, as well as lymphomas and carcinomas. More than one million patients receive it each year. Despite its importance, the bacteria that naturally make doxorubicin do so very inefficiently, forcing manufacturers to rely on costly, multi-step semi-synthetic methods.
Identifying the barriers to production
“We have uncovered several independent factors that limit the formation of doxorubicin,” says Researcher Keith Yamada, PhD, from the University of Turku in Finland, a lead scientist on the study. “By addressing these bottlenecks, we have harnessed rational strain engineering to pave the way for cost-effective manufacturing that can meet growing global demand”.
The research brought together six laboratories across three countries, including the University of Turku in Finland, three groups in the United States, and two in Leiden, the Netherlands.
Working together, the scientists identified three main factors that restrict high-yield production of doxorubicin.
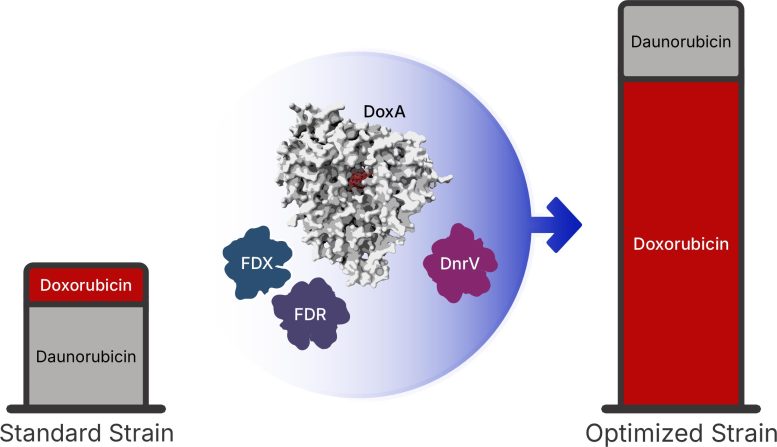
Three discoveries reshape the process
The first finding revealed the enzyme’s required “biological power supply.” The team identified redox partners known as Fdx4 and FdR3, which deliver the electron flow needed to drive the reaction.
Next, the researchers found that a protein called DnrV acts as a drug-binding “molecular sponge.” It sequesters (binds and holds) doxorubicin, preventing the compound from interfering with the enzyme responsible for producing it.
Finally, using X-ray crystallography, the team mapped the enzyme’s structure for the first time. This showed that the drug binds in an unfavorable position inside the enzyme, helping explain why the reaction proceeds so slowly.
From discovery to application
By integrating these insights, the team engineered a bacterial strain capable of producing 180% more doxorubicin than current industrial standards.
To move the technology toward real-world use, the researchers launched the spin-out company Meta-Cells Oy at the University of Turku last year. The company plans to develop these methods for the sustainable production of important antibiotics and anti-cancer drugs. This approach to fully biosynthetic manufacturing could lead to a cleaner and more dependable supply of essential medicines.
Reference: “Metabolic engineering of doxorubicin biosynthesis through P450-redox partner optimization and structural analysis of DoxA” by Arina Koroleva, Erika Artukka, Keith Yamada, Sean A. Newmister, Ralph J. Harte, Hannah Boesger, Mikael Londen, Jacob N. Sanders, Heli Tirkkonen, Matti Kannisto, Rosan C. M. Kuin, Mandy Hulst, Rongbin Wang, Ester Leskinen, Morgane Barillec, Jarmo Niemi, Gilles P. van Wezel, Jacques Neefjes, S. Eric Nybo, Kendall N. Houk, David H. Sherman, Robbert Q. Kim and Mikko Metsä-Ketelä, 4 February 2026, Nature Communications.
DOI: 10.1038/s41467-026-69194-6
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.