
Dietary intake of flavan-3-ols, type of dietary polyphenolics, could help prevent obesity by sympathetic nervous system-induced browning of fat tissue.
In cold conditions, brown adipose tissue (BAT) or brown fat generates heat to keep the body warm. Compared with white adipose tissue, BAT has more mitochondria—subcellular organelles associated with energy production—which allows it to burn calories and produce heat by activating the mitochondrial uncoupling protein 1 (Ucp-1). The stimulation of the sympathetic nervous system (SNS) after cold exposure, exercise, and calorie restriction is well known to induce fat browning. Dietary polyphenols may also activate BAT, causing heat to be dissipated from our bodies. BAT activation and white fat browning are thus both therapeutically significant in the fight against cardiovascular diseases and their comorbidities.
Dietary Flavanols and Their Role in Fat Browning
A group of scientists examined the browning of fat induced by dietary administration of flavan-3-ols (flavanols / FLs), a family of “catechin” containing polyphenols abundant in cocoa, apple, grapeseed, and red wine. In a new study published in the journal Nutrients, the team led by Professor Naomi Osakabe of Graduate School of Engineering and Science, Shibaura Institute of Technology, Japan proved that FLs enhance browning of adipose tissue by activating the SNS. The findings revealed a direct correlation between fat browning and FLs consumption, which could help researchers develop new treatments for obesity-related diseases.
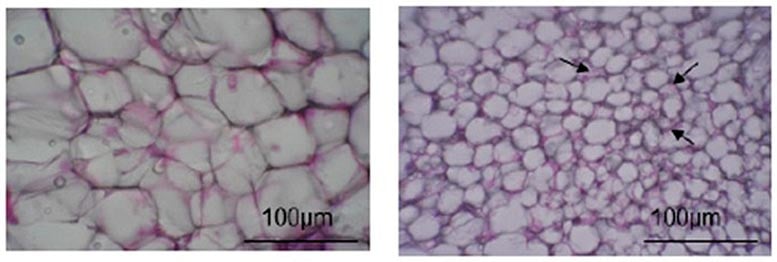
The authors of this study had previously discovered that a single oral dose of FLs caused fat burning and increased skeletal muscle blood flow. Here, they investigated the effects of single and multiple dose administration of FLs in mouse adipose tissue and found that FLs activate fat browning via the SNS, which secretes “catecholamine” neurotransmitters such as adrenaline (AD) and noradrenaline (NA). They fed cocoa-derived FLs to distinct groups of mice in two independent sets of experiments. One group was given a single dose of FLs over the course of 24 hours, and their urine was collected for testing. The other group received repeated doses for 14 days before being dissected for the collection of brown and white fat. All adipose samples were tested for gene and protein markers that indicate fat browning, while the urine samples were tested specifically for AD and NA levels.
Higher concentrations of AD and NA in the urine following a single dose of FL clearly demonstrated SNS activation. Although the use of urine samples to evaluate SNS activation is still controversial in clinical research, it has been validated in stressed rodents. “Oral administration of FLs likely activate the SNS because they are considered stressors in these models,” explains Prof Osakabe.
Long-Term FL Consumption and Its Impact on Fat Browning
The team then used the obtained adipose tissue to investigate the effects of long-term FL treatment. They were thrilled to discover that the white fat of mice who were fed FLs for 14 days eventually turned brown. Some of these cells also had notable structural changes, such as “multilocular phenotype,” and appeared to be smaller than normal cells. Since BAT dissipates heat energy, does long-term FL consumption change the amounts of heat-related proteins? To answer this question, the scientists showed that Ucp-1 levels, as well as other high temperature-linked proteins, increased in mice fed repeated doses of FLs. Browning markers, referred to as “beige markers” in this study, were also abundant in these mice. “All of these proteins work together to induce the development of the BAT phenotype,” exclaims Prof. Osakabe.
The team believes that the results of their study may contribute to the prevention of lifestyle-related diseases. Interestingly, this is not the first time FLs have worked wonders. Improvements in glucose and insulin tolerance have been seen after just one dose of FL-rich food administration. These findings taken together highlight the need of discussing both the acute and chronic aspects of the metabolic responses generated by FLs consumption.
It is evident from this research that the SNS activity in response to FLs intake caused the observed changes in mice fat. “Although the mechanism of adipose browning is not fully understood, it is possible that repeated administration of FLs may produce browning via catecholamines and its receptors,” explains Prof. Osakabe. “Further studies will be required to understand how this process is induced by FL-rich foods,” she concludes.
Reference: “Repeated Oral Administration of Flavan-3-ols Induces Browning in Mice Adipose Tissues through Sympathetic Nerve Activation” by Yuko Ishii, Orie Muta, Tomohiro Teshima, Nayuta Hirasima, Minayu Odaka, Taiki Fushimi, Yasuyuki Fujii and Naomi Osakabe, 24 November 2021, Nutrients.
DOI: 10.3390/nu13124214
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
4 Comments
Interesting.
Examination of Urine and Stools are normal during a Medical Check Ups. However these are cursory.
The optimum BMI(Body Mass Index ) for Humans is Known. Varies with the characteristics of each individual.
To attain this BMI and optimum health, both the Right healthy Diet plus appropriate level of exercise is necessary. So even if we increase the content of Brown Fat and reduce the content of White Belly Fat using appropriate Combination of Food and Exercise, we are only addresing the problem partially.
Why limit the excretory output of the Human / Mammalian Ecosystem to only Urine. Why Not do a detailed analysis of the solid waste excreted (poop) in much greater depth, to get an understanding of the likely problems with the Human and Mamalian Digestive Systems linked to BMI and activity levels? The Other waste we excrete is the Carbon Di Oxide we breathe out which may not be idetical for all humans and Mammalian Species.
The Fact that Life Spans varies for Various Creatures is an indication about the effciciency and effctivenessof various Mammalian Species.
Data Never Lies. People do. Either because of Ignorance or inability to understand the Science .
Best of luck to the Medical Community who fought a valiant battle against a deadly virus with blindfolds on. They do need to be better ared for the next one.
Here is wishing the World a more comprehensive understanding of how to maximise human potential. Both Body and Soul.
Best Wishes for the Medical Revolution afoot to cure ailments both physical and mental. However Soul Cure is not even being addressed.
Views expressed are personal and not binding on anyone.
The info is interesting and warrents more research but ……
Cue the book deals, shelves filled with magic pills and the fleecing of the weak minded.
My thoughts exactly. Not one mention of a FL-rich whole food to eat now. Disappointing. Thankfully there seems to be a growing body of people online who recognize that eating nutritious food can help and they share it. If that wasn’t enough, they share results too. All from eating their veggies, ditching the crap, sourcing high quality local eggs and meat plus going outside to play.
That’s a very very important finding for all the people suffering of this problem and I highly appreciate if you could please send the information I a short and precise way so as to help each and every body who suffers.
Best regards and wishes.