
Scientists say their new insights into how a key brain protein works could help resolve a long-standing question in molecular neuroscience.
Researchers supported by the National Institutes of Health at Johns Hopkins Medicine report that they have uncovered a promising drug target that could help regulate the activity of key brain proteins. By adjusting how these proteins function, future treatments may be able to address psychiatric conditions such as anxiety and schizophrenia, as well as a neurological disorder that affects movement.
The proteins involved are known as delta-type ionotropic glutamate receptors, or GluDs. Scientists have long recognized that GluDs are important for communication between neurons, and mutations in these proteins have been linked to disorders including anxiety and schizophrenia. However, until now, limited understanding of how GluDs actually operate has made it difficult to design therapies that control their activity.
“This class of protein has long been thought to be sitting dormant in the brain,” says Edward Twomey, Ph.D., assistant professor of biophysics and biophysical chemistry at the Johns Hopkins University School of Medicine. “Our findings indicate they are very much active and offer a potential channel to develop new therapies.”
A report on the study’s findings was recently published in Nature.
Structural insight reveals active mechanism
Using cryo-electron microscopy, a highly specialized imaging technique, Twomey and his colleagues were able to examine the structure and behavior of GluDs in detail. Their analysis showed that an ion channel at the center of the GluD protein contains charged particles that help the receptor interact with neurotransmitters (electrical signals that allow brain cells to communicate with one another).
“This process is fundamental for the formation of synapses, the connection point where cells communicate,” says Twomey.
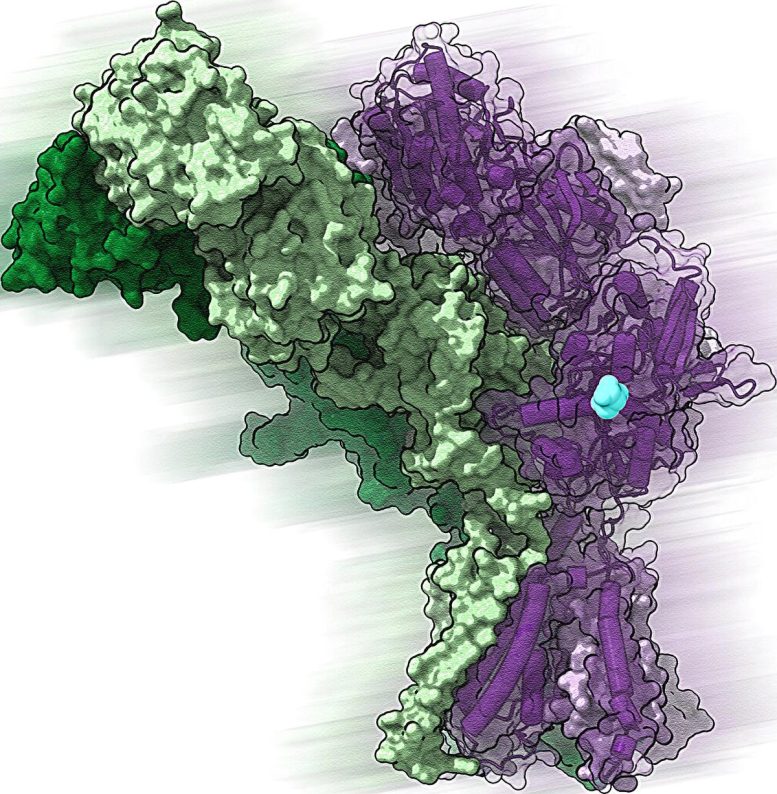
One mechanism, multiple disease paths
The finding could help speed up drug development and discovery in conditions such as cerebellar ataxia, a movement and balance disorder that could result from stroke, head trauma, brain tumors, and certain neurodegenerative conditions. In this disorder, which can also cause memory problems, GluDs become “super-active” even in the absence of electrical signaling. To potentially treat cerebellar ataxia, Twomey says scientists could identify a drug that blocks the hyperactive state in GluDs.
In schizophrenia, where GluDs are known to be less active, Twomey says drugs could potentially dial-up GluD activity.
The findings could also apply to aging and memory loss, in which drugs targeting GluDs could potentially preserve the function of synapses, which play a significant role in the formation of thought and memory.
“Because GluDs directly regulate synapses, we could potentially develop a targeted drug for any condition where synapses malfunction,” Twomey says.
From discovery to therapy development
Up next, Twomey says he is looking to partner with pharmaceutical companies to develop this therapeutic target.
In addition, his team is investigating mutations of GluDs directly implicated in schizophrenia, anxiety, and other psychiatric disorders to better understand disease progression and develop more precise therapeutics.
Reference: “Delta-type glutamate receptors are ligand-gated ion channels” by Haobo Wang, Fairine Ahmed, Jeffrey Khau, Anish Kumar Mondal and Edward C. Twomey, 16 September 2025, Nature.
DOI: 10.1038/s41586-025-09610-x
Disclosure: The Johns Hopkins University has filed a patent for the techniques used to record electrical currents from GluDs.
Funding for this research was provided by the National Institutes of Health (R35GM154904), the Searle Scholars Program and the Diana Helis Henry Medical Research Foundation.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Might these developments help Altxheimers? I’ve got early Altxheimers.