Scientists have found a powerful new way to follow water as it moves around the planet—by tracking subtle “fingerprints” hidden inside its atoms.
Scientists can now follow the path of a single drop of water as it moves through the world.
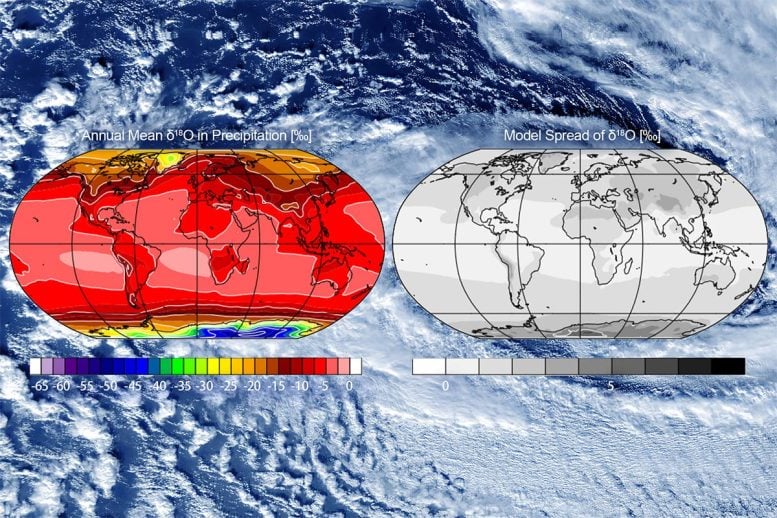
Water is made of hydrogen and oxygen, and some of these atoms naturally occur in heavier forms known as isotopes. As water evaporates, condenses, and travels through the atmosphere, the balance of these isotopes shifts in reliable ways. That pattern works like a signature, letting researchers trace where water has traveled on a global scale.
When combined with hydrological models, these clues help scientists better understand extreme weather such as storms, floods, and droughts, and improve predictions of how climate change may alter weather patterns.
Combining Climate Models for Better Accuracy
Some climate models already include isotopic processes, but accurately simulating the global movement of water remains a major challenge for any single model.
In a study published today (February 10) by Journal of Geophysical Research: Atmospheres, researchers at the Institute of Industrial Science, The University of Tokyo used an approach known as an ensemble, which blends results from multiple models at once. Their ensemble comprised eight climate models that track water isotopes and spanned 45 years from 1979 to 2023. Each model used the same wind and sea-surface temperature data, making it possible to evaluate both individual model behavior and how well the combined average matched real-world observations.
Why Isotopes Reveal Climate Patterns
“Changes in water isotopes reflect shifts in moisture transport, convergence, and large-scale atmospheric circulation. Although we know, at a simple level, that isotopes are affected by temperature, precipitation and altitude, the variability of current model simulations makes it difficult to interpret the results,” said Professor Kei Yoshimura, one of the senior authors of the study, who advised on several of the isotope-enabled climate models participating in the project. “We are delighted that our ensemble mean values capture the isotope patterns observed in global precipitation, vapor, snow, and satellite data much more successfully than any of the individual models.”
Links to Major Climate Systems
When the team analyzed changes over the past 30 years, the ensemble results showed a clear rise in atmospheric water vapor tied to increasing global temperatures. The simulations also revealed strong connections to major climate patterns, including the El Niño-Southern Oscillation, the North Atlantic Oscillation, and the Southern Annular Mode. These large-scale systems influence water availability over multiple years and play a critical role in shaping climate conditions for billions of people around the world.
A World-First Modeling Framework
“Ensembles offer a nuanced modeling approach that reduces divergence between individual models. This approach allows us to separate the effects of how each model represents water cycle processes from differences arising from individual model structures,” said Dr. Hayoung Bong, alumnus of the Institute of Industrial Science, The University of Tokyo, now at NASA Goddard Institute for Space Studies.
By uniting several isotope-enabled climate models into a single framework, the study represents a world-first achievement. The resulting ensemble closely matches observed data, offering a clearer view of how Earth’s water cycle operates.
“Importantly, the research advances our ability to interpret past climate variability and provides a stronger foundation for understanding and predicting how the global water cycle and the weather it shapes will respond to continued global warming,” said Professor Yoshimura.
Reference: “Water Isotope Model Intercomparison Project (WisoMIP): Present-Day Climate” by Hayoung Bong, Allegra N. LeGrande, Sylvia Dee, Jiang Zhu, Alexandre Cauquoin, Richard P. Fiorella, Qinghua Ding, Niels Dutrievoz, Masahiro Tanoue, Michelle Frazer, Mampi Sarkar, Cécile Agosta, Kei Yoshimura, Martin Werner, Atsushi Okazaki, Camille Risi, Hans Christian Steen-Larsen, Mathieu Casado, Sonja Wahl, Jesse Nusbaumer, John Worden, Stephen Good, Adriana Bailey, Matthias Schneider, Stefan Noël, Soumyajit Mandal, Kevin Bowman, Yifan Li and Gavin A. Schmidt, 10 February 2026, Journal of Geophysical Research: Atmospheres.
DOI: 10.1029/2025JD044985
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
“Scientists can now follow the path of a single drop of water as it moves through the world.”
That statement is misleading! It implies that there is a characteristic of water molecules similar to a homing beacon on an aircraft. There is no way to non-destructively sample a single drop of water and then send it back on its way!
“… some of these atoms naturally occur in heavier forms known as isotopes.”
First off, isotopes are NOT just heavier atoms. Isotopes are atoms, all of which have the same number of protons, but differing numbers of neutrons. Some isotopes may have more neutrons (heavier) than the most common, and some may have fewer (lighter); the integer mode is also an isotope. The atomic weight is the average of all naturally-occurring isotopes.
“We are delighted that our ensemble mean values capture the isotope patterns observed in global precipitation, vapor, snow, and satellite data much more successfully than any of the individual models.”
The unstated assumption is that all model ensembles will provide more accurate results than one or two models because outliers are suppressed by an averaged ensemble because the probability of an outlier is usually low. However, that assumption is only true if ALL the models regress about the correct results. That is, the results will be randomly distributed (symmetrical) about the correct value. However, a weakness in using model ensembles is that there can be a bias in the sampling or calculation, as appears to be the case with global temperature models that tend to run warm compared to observations. Furthermore, logically, there can only be one best model and averaging that with all the inferior models gives an answer that is different from the best model and possibly different from the true value. Thus, one is trading a reduction in the probability of an anomalously large error (a single outlier) by reducing the precision and increasing the probability that the ensemble average will not be as good as the best member of the ensemble.
I’m encouraged that the authors think that their ensemble approach improves the estimate of the isotopic fractionation of atmospheric water. However, I’m skeptical that ensembles will always be symmetrical and that the mean will always fall close to the true value. Neither this press release nor the available abstract speak to the probability distribution of the 8 model samples, which could just coincidentally be producing better results than any single model.