Scientists at Georgia State University have used CRISPR gene editing to restore an ancient enzyme humans lost millions of years ago potentially reversing the buildup of uric acid that causes gout.
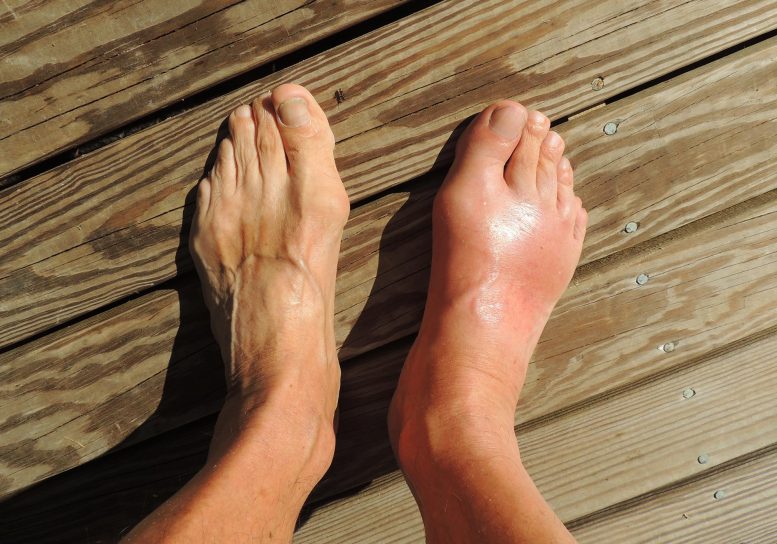
Gout is an ancient form of arthritis caused by the buildup of sharp crystals in the joints, leading to swelling and intense pain. Now, scientists at Georgia State University believe they may have uncovered an ancient method to combat it.
In a study published in Scientific Reports, the research team used CRISPR gene-editing technology to restore a gene that humans lost millions of years ago, which resulted in lower levels of uric acid—the compound responsible for gout and several other health issues.
The key lies in an enzyme called uricase, which most other animals still possess.
Uricase helps the body break down uric acid, a waste product that forms naturally in the blood. When too much of it accumulates, the excess can crystallize in joints and kidneys, causing gout, kidney stones, and related health problems.
Humans and other apes lost the uricase gene about 20 to 29 million years ago. Some researchers believe this genetic loss once served a purpose. For example, Dr. Richard Johnson of the University of Colorado has suggested that higher uric acid levels may have helped early primates convert fruit sugar into fat, offering a survival edge during periods of food scarcity, as described in a paper Seminars in Nephrology.
Turning an Evolutionary Quirk Into a Medical Solution
Still, what once helped our ancestors survive now contributes to modern diseases, and that’s what Eric Gaucher, a biology professor at Georgia State, and his team set out to challenge.
“Without uricase, humans are left vulnerable,” said Gaucher, the study’s co-author. “We wanted to see what would happen if we reactivated the broken gene.”
Working with postdoctoral researcher Lais de Lima Balico, Gaucher used CRISPR-Cas9 — a gene-editing tool often called molecular scissors — to put a reconstructed ancient uricase gene into human liver cells so they could see how the enzyme worked.
The results were dramatic: Uric acid dropped and fructose-driven fat buildup in liver cells was prevented. But results in isolated cells aren’t always enough, so the team pushed the experiment further.
To see if the gene would behave the same way in more complex conditions, the team moved from simple liver cells to 3D liver spheroids. These miniature lab-grown tissues mimic how organs work in the body. The revived uricase gene lowered uric acid there, too. The enzyme also found its way to peroxisomes — tiny compartments inside cells where uricase normally does its job. That finding suggests the therapy could function safely in living systems, not just in isolated cells.
“By reactivating uricase in human liver cells, we lowered uric acid and stopped the cells from turning excess fructose into triglycerides — the fats that build up in the liver,” Gaucher said.
Beyond Gout: A Wider Health Impact
This discovery matters far beyond gout. High uric acid, also called hyperuricemia, is tied to a range of modern health problems, not just painful joints. Studies link it to hypertension and cardiovascular disease, among others — with risks that researchers such as Johnson have compared to high cholesterol, according to research in the journal Hypertension.
Those risks aren’t just theoretical. They show up clearly in patient data.
About a quarter to half of patients with high blood pressure also have elevated uric acid, and in new hypertension cases, the overlap jumps to 90 percent, according to the study.
“Hyperuricemia is a dangerous condition,” Gaucher said. “By lowering uric acid, we could potentially prevent multiple diseases at once.”
Current gout treatments don’t work for everyone, and some patients react badly to lab-made uricase therapies. A CRISPR-based approach could sidestep those problems by restoring uricase inside liver cells.
“Our genome-editing approach could allow patients to live gout-free lives and potentially prevent fatty liver disease,” Gaucher said.
Next come animal studies and, if results hold, human trials. Potential delivery options range from direct injections to returning lab-modified liver cells to patients. Another option is lipid nanoparticles — the same technology used in some COVID-19 vaccines.
If the approach proves safe, Gaucher said it could transform treatment for gout and related metabolic diseases. But hurdles remain.
“Genome-editing still faces substantial safety concerns,” he said. “Once those are addressed, society will be faced with contentious ethical discussions about who should and should not have access.”
Reference: “Genomic insertion of ancestral uricase into human liver cells to determine metabolic consequences of pseudogenization” by Lais de Lima Balico, and Eric A. Gaucher, 18 July 2025, Scientific Reports.
DOI: 10.1038/s41598-025-10551-8
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
I currently are suffer from gout in right ankle and foot.
I the find this research very interesting.
As do I. The idea of living a life free of gout is a dream come true. Hopefully, in my lifetime, the therapy will be available to all. Otherwise it’s just a cruel joke perpetrated by big pharma.