
Scientists have discovered a mercury-free method to isolate lithium-6, a vital ingredient in nuclear fusion fuel.
The breakthrough emerged by accident during water purification research and uses a material called zeta-vanadium oxide to selectively trap lithium-6 ions. The new method avoids the toxic COLEX process and has shown promising enrichment results in lab tests. Researchers now aim to scale the process to help power the future of clean energy.
Mercury-Free Method Unlocks Lithium-6 for Fusion Fuel
Lithium-6 is a key ingredient for producing fuel used in nuclear fusion, but separating it from the far more abundant lithium-7 typically requires liquid mercury, a highly toxic substance. Now, scientists have developed a mercury-free method that can isolate lithium-6 just as effectively as the traditional approach. Their findings were published on March 20 in the journal Chem, published by Cell Press.
“This is a step towards addressing a major roadblock to nuclear energy,” says chemist and senior author Sarbajit Banerjee of ETH Zürich and Texas A&M University. “Lithium-6 is a critical material for the renaissance of nuclear energy, and this method could represent a viable approach to isotope separation.”
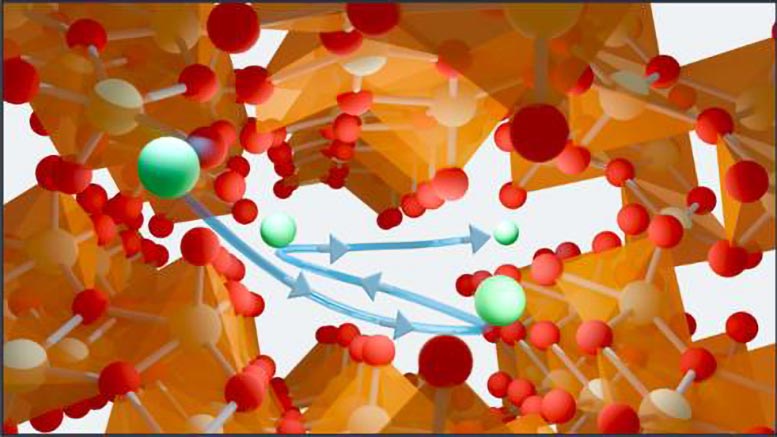
Toxic Legacy of COLEX and the Lithium-6 Shortage
The standard technique to isolate lithium-6, known as the COLEX process, uses liquid mercury and has been banned in the U.S. since 1963 due to environmental and health concerns. Since then, U.S. research has relied on a limited stockpile of lithium-6 maintained at Oak Ridge National Laboratory in Tennessee. Finding a safer, scalable method to isolate lithium-6 is essential for advancing nuclear fusion as a clean energy source.
Interestingly, the new method was discovered by accident. The researchers were developing filtration membranes to clean “produced water,” a type of wastewater brought to the surface during oil and gas drilling, when they noticed the membranes were capturing unusually high amounts of lithium. That unexpected result led them to investigate further, ultimately uncovering a new way to separate lithium-6 without mercury.
A Hunch Leads to Isotope Selectivity Testing
“We saw that we could extract lithium quite selectively given that there was a lot more salt than lithium present in the water,” says Banerjee. “That led us to wonder whether this material might also have some selectivity for the 6-lithium isotope.”
The membrane’s lithium-binding properties are due to a material called zeta-vanadium oxide (ζ-V2O5), a lab-synthesized inorganic compound that contains a framework of tunnels running in a single dimension.
“Zeta-V2O5 has some pretty incredible properties—it’s an amazing battery material, and now we’re finding that it can trap lithium very selectively, even with isotopic selectivity,” says Banerjee.
Electrochemical Cell Confirms Lithium-6 Capture
To test whether the material could separate lithium-6 from lithium-7, the team set up an electrochemical cell with a zeta-V2O5 cathode. When they pumped an aqueous solution containing lithium ions through the cell while applying a voltage, the positively charged lithium ions were drawn towards the negatively charged zeta-V2O5 matrix and into its tunnels. Because lithium-6 and lithium-7 ions move differently, the zeta-V2O5 tunnels preferentially captured lithium-6 ions while the more mobile lithium-7 ions escaped capture.
“Lithium-6 ions stick a lot stronger to the tunnels, which is the mechanism of selectivity,” says co-first author Andrew Ezazi of Texas A&M. “If you think of the bonds between V2O5 and lithium as a spring, you can imagine that lithium-7 is heavier and more likely to break that bond, whereas lithium-6, because it’s lighter, reverberates less and makes a tighter bond.”
As lithium ions are integrated into the zeta-V2O5, the compound gradually changes color from bright yellow to dark olive green, which enables the degree of lithium isolation to be easily monitored.
Reaching Fusion-Grade Lithium Without Mercury
The team shows that a single electrochemical cycle enriched lithium-6 by 5.7%. To obtain fusion-grade lithium, which requires a minimum of 30% lithium-6, the process needs to be repeated 25 times, and 90% lithium-6 can be obtained in about 45 sequential cycles.
“This level of enrichment is very competitive with the COLEX process, without the mercury,” says Ezazi.
Toward Scalable, Cost-Effective Fusion Fuel
“Of course, we’re not doing industrial production yet, and there are some engineering problems to overcome in terms of how to design the flow loop, but within a bunch of flow cycles, you can get fusion-grade lithium for quite cheap,” says Banerjee.
The researchers say that their results suggest that materials like zeta-V2O5 could be used to isolate other substances, for example, to separate radioactive from non-radioactive isotopes.
Now, the team is taking steps to scale their method up to an industrial level.
“I think there’s a lot of interest in nuclear fusion as the ultimate solution for clean energy,” says Banerjee. “We’re hoping to get some support to build this into a practicable solution.”
Reference: “Electrochemical 6Li isotope enrichment based on selective insertion in 1D tunnel-structured V2O5” by J. Luis Carrillo, Andrew A. Ezazi, Saul Perez-Beltran, Carlos A. Larriuz, Harris Kohl, Jaime A. Ayala, Arnab Maji, Stanislav Verkhoturov, Mohammed Al-Hashimi, Hassan Bazzi, Conan Weiland, Cherno Jaye, Daniel A. Fischer, Lucia Zuin, Jian Wang and Sarbajit Banerjee, 20 March 2025, Chem.
DOI: 10.1016/j.chempr.2025.102486
This research was supported by the National Science Foundation, Texas A&M, the Canada Foundation for Innovation, the Natural Sciences and Engineering Research Council of Canada, the Canadian Institutes of Health Research, the National Research Council Canada, the Canadian Institutes of Health Research, the Government of Saskatchewan, the University of Saskatchewan, and the Qatar Research, Development and Innovation Council.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
4 Comments
Aren’t the filter material toxic with Lithium 7?
My understanding there is more lithium 7 than the useful Lithium 6.
So where does that go and is it really clean energy?
Vanadium is less toxic than mercury, it’s also far easier to handle being a room-temperature solid.
They don’t say what they do with the Lithium-7, but I’m guessing there are uses for it somewhere and it just goes back into the supply chain.
Lithium is in demand for making rechargeable batteries – that’s one of the key uses for it.
I enjoyed this. It was understandable to me even though I have no specialized knowledge of this subject.