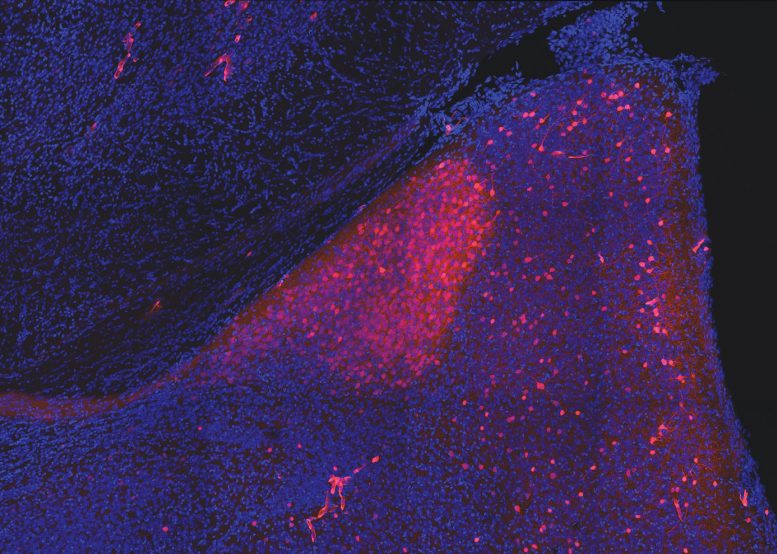
Columbia-led study in mice maps brain circuitry that enables a sex pheromone named for Jane Austen character to alter brain in mouse courtship.
The infamously aloof Mr. Darcy had a hard time attracting members of the opposite sex in Jane Austen’s Pride and Prejudice. But the same cannot be said for a sex pheromone named for him, called darcin. In a new study, a Columbia University-led team of researchers has now uncovered the process by which this protein takes hold in the brains of female mice, giving cells in the brain’s emotion center the power to assess the mouse’s sexual readiness and help her select a mate.
These findings, published in the February 6, 2020 issue of Nature, illustrate the power of a single protein to change the brain and drive behavior. They also demonstrate how a cluster of cells in one brain area integrates information from the outside world with the animal’s own internal state.
“Pheromones act as powerful scent messages to signal the presence of danger, food or prospective mates,” said Ebru Demir, Ph.D., the paper’s first author. “With today’s study, we’ve mapped the route that the pheromone darcin takes from the nose to the brain, bringing much-needed understanding to the mechanisms by which animals use scents to communicate,” added Dr. Demir, who is an associate research scientist in the laboratory of Nobel Laureate Richard Axel, MD, at Columbia’s Mortimer B. Zuckerman Mind Brain Behavior Institute.
While the existence of pheromones in humans is controversial, rodents and many other animals rely on pheromones as a way to signal everything from potential danger to a willingness to mate.
Darcin is one such pheromone, discovered in 2010 by Robert Beynon, Ph.D., and Jane Hurst, Ph.D., and their team at the University of Liverpool. Dr. Hurst and her colleagues found that male mice release darcin in their urine to mark their territory and to initiate courtship displays. Sniffing a male’s darcin helps a female to both identify him and decide whether to mate with him. This entire process is initiated in a biologically unusual way.
Normally, mice make sense of odors using olfactory receptors in the nose. These specialized proteins send information about a scent to a designated location in the brain for further processing. Dr. Axel, who is codirector at Columbia’s Zuckerman Institute, received the 2004 Nobel Prize in Physiology or Medicine jointly with Linda Buck, Ph.D., for their work identifying the genes that encode these receptors.
Pheromones, such as darcin, are processed somewhat differently. They interact with a second, parallel olfactory system, which exists in animals like mice but not in people.
“Unlike people, mice have essentially two functional noses,” said Dr. Demir. “The first nose works like ours: processing scents such as the stinky odor particles found in urine. But a second system, called the vomernasal nose, evolved specifically to perceive pheromones like darcin.”
For today’s study, the research team, which also included Dr. Hurst, Dr. Beynon, and co-senior author Adam Kepecs, Ph.D., of Cold Spring Harbor Laboratory, first exposed female mice to darcin-scented urine and monitored their behavior. Nearly all of the female mice showed an immediate attraction to darcin. Then, after about 50 minutes, some females began leaving their own urinary scent markings. They also started to sing, at ultrasonic frequencies too high for the human ear to hear. Both of these behaviors are an indicator of increased sexual drive.
Not all females performed these displays. Lactating mothers, for example, appeared to largely ignore the darcin-scented areas after an initial sniff of interest.
The reason for this difference, the scientists proposed, may lie in a brain region called the medial amygdala. The research team identified a subset of brain cells, or neurons, in this brain area, called nNOS neurons, that switched on in the presence of darcin.
“By artificially activating those neurons, we could simulate the animals’ response to darcin and elicit the same behaviors,” said Dr. Demir. “When we silenced these neurons, the animal lost interest in darcin entirely.”
The neurons’ location in the medial amygdala was particularly intriguing. This brain area is generally associated with hardwired emotional responses, such as fear or anger. In the case of the darcin pheromone, though, the medial amygdala may serve another role.
“Our results suggest that nNOS neurons in the medial amygdala do not simply pass along information about darcin,” said Dr. Demir. “These neurons seem to be integrating sensory information about the pheromone with the internal state of the animal, such as whether she is a lactating mother and therefore not interested in mating.”
Going forward, the research team plans to delve deeper into the neural circuitry that responds to pheromones and how changes to that circuitry drive behavior. They also hope their findings will serve to update how pheromones are defined.
“Pheromones have long been associated with an innate, immediate behavioral response, but here we have shown that darcin can elicit complex behaviors that are dependent on the internal state of the animal,” said Dr. Demir. “As we continue our investigations, it’s possible that other pheromones may also act on the brain in similarly unexpected and complex ways.”
This paper is titled “The pheromone darcin drives a circuit for innate and reinforced behaviours.” Additional contributors include Kenneth Li, Natasha Bobrowski-Khoury, and Joshua Sanders, Ph.D.
Reference: “The pheromone darcin drives a circuit for innate and reinforced behaviours” by Ebru Demir, Kenneth Li, Natasha Bobrowski-Khoury, Joshua I. Sanders, Robert J. Beynon, Jane L. Hurst, Adam Kepecs and Richard Axel, 29 January 2020, Nature.
DOI: 10.1038/s41586-020-1967-8
This research was supported by the Howard Hughes Medical Institute, the Biotechnology and Biological Sciences Research Council and the Robert E. Leet and Clara Guthrie Patterson Trust Fellowship.
The authors report no financial or other conflicts of interest.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.