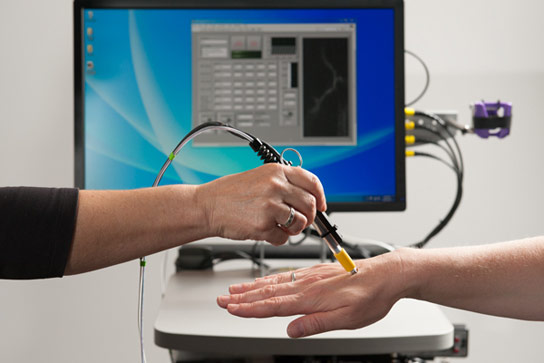
Stanford scientists are developing a mini-microscope to noninvasively detect circulating tumor cells, allowing for earlier interventions.
One of the cruelest truths about cancer is that even after you beat the disease, it can still come back to kill you. A tumor growing in the prostate gland, breast, or any other organ can shed cancerous cells into the blood. These cancerous seeds travel the body and can take root nearly anywhere, growing into a new cancer threat even after the initial cancer is treated.
The rule of thumb with cancer is that the earlier you can detect the disease, the more effective the treatment, and hence better potential outcomes.
Currently, doctors draw a patient’s blood and analyze it using special antibodies to detect the presence of the seeds, called circulating tumor cells (CTCs). This works well if CTCs are present in large numbers, but may fail to detect smaller numbers released by earlier tumors.
Now, a team of engineers, scientists, and doctors from Stanford is developing a mini-microscope that might be able to noninvasively detect the CTCs earlier than ever, allowing for earlier interventions.
“There has been a huge push to increase sensitivity,” said Bonnie King, an instructor at Stanford School of Medicine. “We suspect that CTCs often circulate in numbers below our current threshold of detectability.”
A major advantage of the microscopic technique, King said, is the ability to screen much larger volumes of blood, rather than just a small vial collected from a patient. This will be done using a method called in vivo flow cytometry – a laser-based technology for counting cells in a live subject.
Christopher Contag, a professor of pediatrics at the School of Medicine, envisions that a doctor would inject a patient with a dye that will cause the CTCs to fluoresce. The doctor would then use the pen-size microscope to focus a low-power laser light on a blood vessel just a few hair-widths below the patient’s skin.
As the dyed cancer cells pass through the laser, the light excites them and causes them to stand out from normal cells. The microscope registers each of these cells and a computer logs each observation. The improved sensitivity of the technology and the ability to noninvasively scan blood for long periods will help create a fuller picture of the number of CTCs in a person’s body.
“At present, we will not screen all of a person’s blood [with the microscope], but we are aiming to increase the amount of blood screened compared to a 7-milliliter blood draw,” Contag said.
The work is a collaborative effort of Olav Solgaard, a professor of electrical engineering; Geoffrey Gurtner, a professor of surgery; and Michael Clarke, a professor of oncology. It began last fall, when the proposed project was awarded a seed grant by Stanford’s Bio-X Interdisciplinary Initiatives Program.
Every two years, Bio-X provides funding to interdisciplinary projects that could lead to innovative improvements in human health. Since 2000, Bio-X seed grants have funded 141 research collaborations connecting hundreds of faculty. The proof-of-concept projects have produced hundreds of scientific publications, dozens of patents, and more than a tenfold return on research funds to Stanford.
To date, the blood-scan group has focused on developing the method in mice, taking advantage of the thin transparent tissue of the ear to image fluorescent cells traversing the small blood vessels below the skin.
Soon the researchers will move the microscope to a clinical setting to conduct a proof-of-principle test of the technique in humans. Gurtner is currently conducting a clinical trial to evaluate the FDA-approved green dye for defining skin vasculature during post-mastectomy breast reconstruction surgeries. The researchers are piggybacking on this trial to test the miniature microscope’s ability to detect blood vessels and circulating cells.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.