Human skin cells reprogrammed into placental stem cells shows how COVID infects placenta — and how it can be stopped.
In a landmark study published today (July 13) in the journal Nature Cell Biology, Australian researchers, led by Professor Jose Polo from Monash University and the University of Adelaide and University of Melbourne’s Professor Kanta Subbarao from the Peter Doherty Institute for Infection and Immunity (Doherty Institute), have revealed how COVID-19 can infect the human placenta.
Research has shown that COVID-19 infections during pregnancy may lead to adverse outcomes, but little is known about the mechanisms behind the effects of SARS-CoV-2 infection in pregnancy.
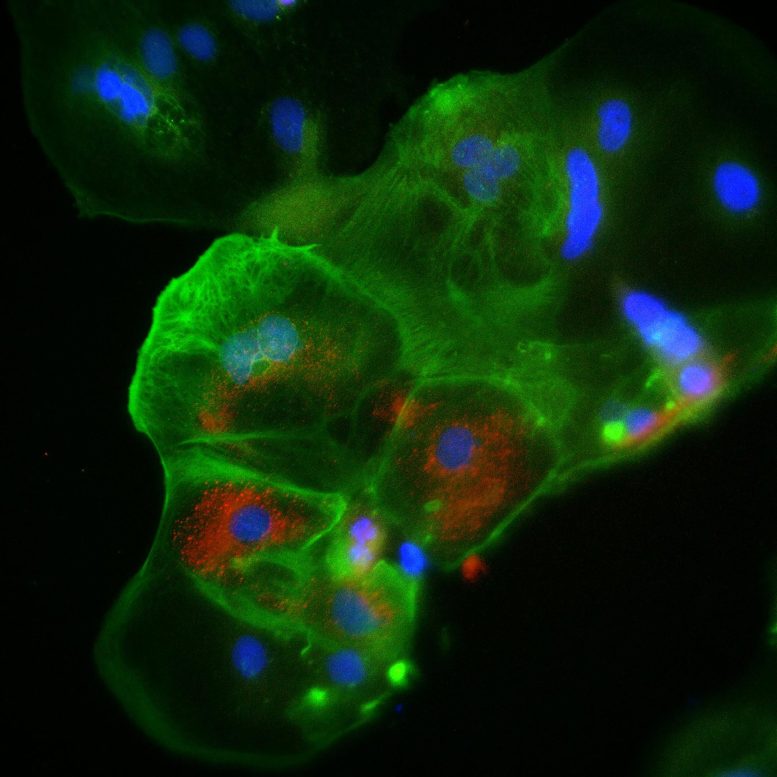
The Australian research team grew placenta tissue in the lab, using a state-of-the-art method developed by Professor Polo and colleagues where human skin cells are “reprogrammed” into trophoblast stem cells (the cells that help a developing embryo attach to the wall of the uterus, forming part of the placenta). They found that ACE2, a protein that acts as the doorway for SARS-CoV-2 to enter organs such as the lung, is present in specific placental cells, like syncytiotrophoblasts (ST cells).
Importantly, ST cells were susceptible to the virus – a major finding as these placental cells produce the key hormone for maintaining pregnancy (hCG).
Dr. Joseph Chen, a stem cell biologist at Monash University and co-first author of the report, said this discovery explains several clinical reports indicating inflammation of the placenta due to COVID-19.
“We observed that SARS-CoV-2 infection led to a significant reduction in the survival and differentiation of ST cells, which in turn resulted in lower production of hCG,” he said. “It suggests that this is how COVID-19 could impact pregnancy, though further investigations are needed.”

Virologist at Doherty Institute and co-first author of the study Dr Jessica Neil said, “our team also discovered that anti-ACE2 antibodies and antiviral drugs were effective in preventing SARS-CoV-2 infection and restoring normal ST differentiation and function”.
Professor Subbarao said that this study is a significant advance for the broader understanding of viral infections in pregnancy.
“Our study provides valuable insights into the link between SARS-CoV-2 infection and placenta pathology. This is a game changer as we are now equipped to explore how the early placenta may be affected by other viruses as well,” she said.
Professor Polo emphasized the importance of the research in establishing a platform to study early placental cell types.
“This study not only helps us to understand what happens when the placenta is infected with the COVID-19 virus during pregnancy, it also means we have established a broader, scalable and tractable platform to study early placental cell types,” he said.
Reference: “A placental model of SARS-CoV-2 infection reveals ACE2-dependent susceptibility and differentiation impairment in syncytiotrophoblasts” by J. Chen, J. A. Neil, J. P. Tan, R. Rudraraju, M. Mohenska, Y. B. Y. Sun, E. Walters, N. G. Bediaga, G. Sun, Y. Zhou, Y. Li, D. Drew, P. Pymm, W. H. Tham, F. J. Rossello, G. Nie, X. Liu, K. Subbarao and J. M. Polo, 13 July 2023, Nature Cell Biology.
DOI: 10.1038/s41556-023-01182-0
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.