Inspired by how bones heal naturally, researchers have engineered a laser-structured hydrogel that could one day replace conventional bone implants.
Bones fractured in a (skiing) accident often mend without medical intervention. However, when a break is especially severe, or a bone tumor must be surgically removed, doctors may place an implant to stabilize the area and help the bone knit back together.
These implants are commonly made from the patient’s own bone, referred to as autografts, or from metal or ceramic materials. One major limitation of many current treatments is that harvesting autografts requires a second surgical procedure. Metal implants also present challenges. Because they are typically much stiffer than natural bone, they can loosen over time and reduce long-term stability.
Taking biology into account
Bone is not simply a hard, lifeless structure. It is a living organ filled with an intricate system of tiny channels and hollow spaces.
“For proper healing, it is vital that biology is incorporated into the repair process,” says Xiao-Hua Qin, Professor of Biomaterials Engineering at ETH Zurich. For healing to succeed, different types of cells must first move into the implant and establish themselves there before new bone tissue can form.
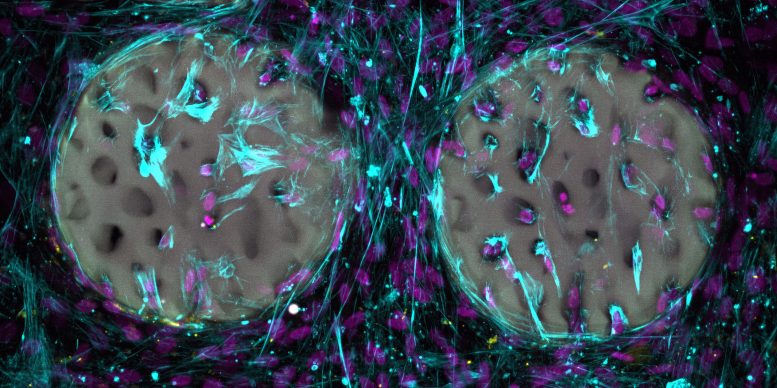
With this in mind, Qin and his team, working alongside ETH Professor Ralph Müller, developed a new type of hydrogel designed for future implant use. The material has a soft, jelly-like consistency and slowly dissolves inside the body. It could potentially be customized for individual patients. The research describing this advance was recently published in the journal Advanced Materials.
Healing begins with soft material
According to the researchers, bone repair naturally starts with a soft scaffold. Shortly after a fracture occurs, the body forms a hematoma, or bruise, at the injury site. This permeable structure allows immune cells and repair cells to migrate into the area while also enabling nutrients to flow in. A network of fibrin holds these cells together. Over time, this flexible framework is gradually replaced by solid, mineralized bone.
The newly developed hydrogel is designed to replicate this early stage of healing. It consists of 97 percent water and 3 percent biocompatible polymer. To transform it from liquid to solid, the team incorporated two specialized molecules. One connects the polymer chains, while the other activates the linking process when exposed to light.
Wanwan Qiu, a former doctoral student of Qin and Müller, created the linking molecule specifically for this purpose. “It enables rapid structuring of hydrogels in the sub-micrometer range,” she says. When laser pulses of a specific wavelength strike the material, the polymer chains immediately bond in the exposed regions. The illuminated sections solidify at once, while the remaining material can later be rinsed away.
Jelly can be set at world-record speed
Using a focused laser beam, the researchers can shape the hydrogel into nearly any desired form with exceptional precision. The smallest features they can produce measure just 500 nanometers.
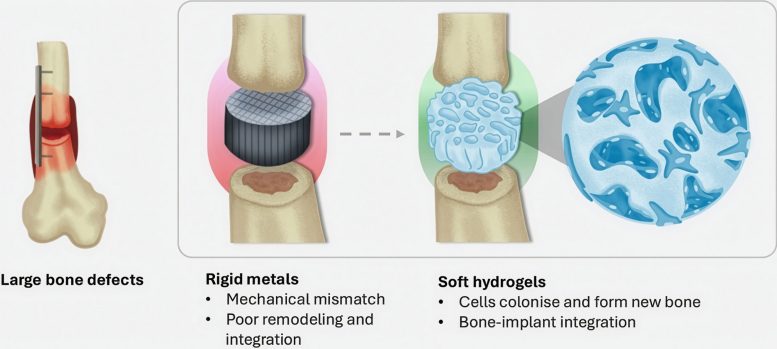
“Hydrogels resemble jelly, making them difficult to shape,” says ETH Professor Qin. “With our newly developed connecting molecule, we can now not only structure the hydrogel in a stable and extremely fine manner but also produce it at high writing speeds of up to 400 millimeters per second. That’s a new world record.”
This combination of speed and fine detail allows the team to construct delicate internal patterns that closely mimic natural bone.
Structures in the nanometre range
In their study, the researchers created complex, structured hydrogels that resemble real bone and feature a fine network of bone trabeculae. They used medical imaging as a template.
Even healthy natural bone is crisscrossed by a fine network of channels that are only nanometers thick and filled with fluid. “A piece of bone the size of a dice contains 74 kilometers of tunnels,” says Qin. By way of comparison, the longest railway tunnel in the world, the Gotthard Base Tunnel, measures 54 kilometers.
Material is biocompatible
To date, the material has been tested only in laboratory settings. In these in vitro experiments, bone-forming cells quickly settled into the structured hydrogel and began producing collagen, a crucial building block of bone tissue. The tests also showed that the material is biocompatible and does not harm these cells. The team has patented the underlying material and intends to make it available to the medical industry.
Ultimately, Qin hopes the hydrogel-based implant will be used in clinical practice to treat bone injuries. Additional research is still required. He is preparing to begin animal studies in partnership with the AO Research Institute Davos. These studies will investigate whether the material encourages bone-forming cells to migrate in living organisms and whether it can restore bone strength over time.
Reference: “A Water-Soluble PVA Macrothiol Enables Two-Photon Microfabrication of Cell-Interactive Hydrogel Structures at 400 mm s−1” by Wanwan Qiu, Margherita Bernero, Muja Emilie Ye, Xianjun Yang, Philipp Fisch, Ralph Müller and Xiao-Hua Qin, 8 January 2026, Advanced Materials.
DOI: 10.1002/adma.202510834
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Could this work on 15 years old Golden Retrievers with bone on bone arthritis and an improperly set knee ligament tear?