Using QCM-D, scientists observed how hydration structures impact ion transport and storage in layered materials. This discovery links charge density to ion storage potential and offers pathways to enhance energy storage and ion separation.
Studying the relationship between the arrangement of water molecules incorporated into layered materials like clays and the arrangement of ions within these materials has been a difficult experiment to conduct.
However, researchers have now succeeded in observing these interactions for the first time by utilizing a technique commonly used for measuring extremely small masses and molecular interactions at the nanoscale.
The findings were recently published in the journal Nature Communications.
Many materials take a layered form at the microscopic or nano-scale. When dry, clays for example resemble a series of sheets stacked upon each other. When such layered materials encounter water, however, that water can be confined and integrated into the gaps or holes—or, more accurately, the ‘pores’—between layers.
Such ‘hydration’ can also occur when water molecules or their constituent elements, notably a hydroxide ion (a negatively charged ion combining a single oxygen and single hydrogen atom) are integrated into the crystalline structure of the material. This type of material, a ‘hydrate’, is not necessarily ‘wet’ even though water is now part of it. Hydration can also substantially change the original material’s structure and properties.
Nanoconfinement and Ion Storage Potential
In this ‘nanoconfinement’, the hydration structures—how water molecules or their constituent elements arrange themselves—determine the ability of the original material to store ions (positively or negatively charged atoms or groups of atoms).
This storage of water or charge means that such layered materials, from conventional clays to layered metal oxides—and, crucially, their interactions with water—have widespread applications, from water purification to energy storage.
However, studying the interplay between this hydration structure and the configuration of ions in the ion storage mechanism of such layered materials has proven to be a great challenge. And efforts at analyzing how these hydration structures change over the course of any movement of these ions (‘ion transport’) are even more difficult.
Recent research has shown that such water structures and interactions with the layered materials play an important role in giving the latter their high ion-storage capacities, all of which in turn depend upon how flexible the layers that host the water are. In the space between layers, any pores that are not filled with ions get filled with water molecules instead, helping to stabilize the layered structure.
“Put another way, the water structures are sensitive to how the interlayer ions are structured,” said Katsuya Teshima, corresponding author of the study and a materials chemist with the Research Initiative for Supra-Materials at Shinshu University. “And while this ion configuration in many different crystal structures controls how many ions can be stored, such configurations until now had rarely been systematically investigated.”
Quartz Crystal Microbalance
So Teshima’s group looked to ‘quartz crystal microbalance with energy dissipation monitoring’ (QCM-D) to assist with their theoretical calculations. QCM-D is essentially an instrument that works like a balance scale that can measure extremely tiny masses and molecular interactions at the nano level. The technique can also measure tiny changes in energy loss.
The researchers used QCM-D to demonstrate for the first time that the change in the structure of water molecules confined in the nano-space of layered materials can be experimentally observed.
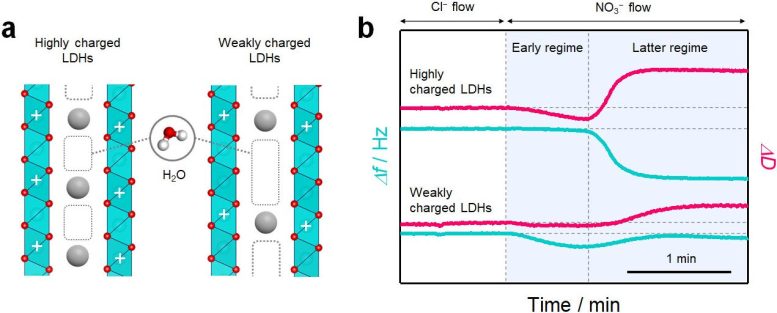
They did this by measuring the “hardness” of the materials. They investigated the layered double hydroxides (LDHs) of a class of negatively charged clay. They found that the hydration structures were associated with the hardening of the LDHs when any ion exchange reaction happens (a swapping of one kind of ion with a different type of ion but with the same change).
“In other words, any change in ion interaction originates with the change in the hydration structure that occurs when ions are incorporated into the nano-space,” added Tomohito Sudare, a collaborator on the study now with the University of Tokyo.
Hydration Structures and Ion Storage Capacity
In addition, the researchers found that the hydration structure is highly dependent on the charge density (the amount of charge per unit of volume) of the layered material. This in turn is largely what governs the ion storage capacity.
The researchers now hope to apply these measurement methods together with the knowledge of the hydration structure of ions to devise new techniques for improving the ion-storage capability of layered materials, potentially opening new avenues for ion separation and sustainable energy storage.
Reference: “Critical role of water structure around interlayer ions for ion storage in layered double hydroxides” by Tomohito Sudare, Takuro Yamaguchi, Mizuki Ueda, Hiromasa Shiiba, Hideki Tanaka, Mongkol Tipplook, Fumitaka Hayashi and Katsuya Teshima, 28 October 2022, Nature Communications.
DOI: 10.1038/s41467-022-34124-9
The study was funded by the Environmental Restoration and Conservation Agency, the Japan Society for the Promotion of Science, and the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.