Early tests at UChicago show promise for treating resistant tumors with unique approach.
One of the most promising avenues in treating cancer is to restore our immune system’s ability to recognize and attack cancerous cells. A team of University of Chicago chemists and biologists developed a tiny device that can locate tumor cells and force them to reveal themselves to patrolling immune cells. In tests with mice, this resulted in tumor regression.
“When it comes to drug delivery, the problem, as molecular biologist Inder Verma put it, is delivery, delivery, and delivery,” explained Yamuna Krishnan, a professor in the Department of Chemistry and an author of the study. “These DNA nanodevices now make drug delivery hyperspecific, allowing us to think of ways to treat cancer without killing the cell that the therapeutic is delivered to.”
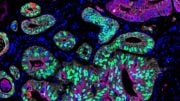
The focus of these nanodevices is a particular type of cell known as tumor-associated macrophages, or TAMs. Macrophages are a type of immune cell that normally is supposed to recognize and remove microbes, cellular debris, and other foreign substances from cells; but if something goes wrong with them, they can become a key part of cancerous tumors. TAMs can comprise up to 50% of tumor mass in triple-negative breast cancer.
However, “despite the high abundance of TAMs in solid tumors, mechanisms underlying their impact on tumor development and therapeutic strategies to target them are incompletely understood,” said study co-author Lev Becker, associate professor in the Ben May Department for Cancer Research.
The importance of these TAMs goes back to how the immune system recognizes cancerous cells. There is a subpopulation of immune cells called CD8+ T cells that are critical in recognizing and killing cancerous cells. These CD8+ T cells can be activated against threats by binding to molecular structures called “antigens” on the surface of cancerous macrophages. This strategy goes awry, however, when TAMs don’t present antigens, so there is nothing for the T-cells to recognize.
Becker’s group found that TAMs harbored a high level of a kind of enzyme called cysteine proteases. They knew these particular enzymes live in lysosomes, which work as the “stomach” of the cell, so Becker’s insight was that they might be “over-digesting” tumor antigens –thereby concealing cancerous cells from patrolling CD8+ T cells.
To test this idea, Becker’s group needed to prove that the problem really did lie in the lysosomes eating away at the antigens. So they used mice whose macrophages lacked a protein that regulates lysosomal enzyme levels and activity. They found that indeed, the lysosomes in the TAMs of these mice weren’t destroying antigens as much. This ultimately allowed CD8+ T cells to “see” and attack the tumor.
Next, they needed to figure out a way to target this process therapeutically.
“Our intent was not to kill the target cells, but rather reprogram them and change their character.”
— Prof. Yamuna Krishnan
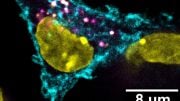
Meanwhile, Krishnan, an expert in DNA nanotechnology, had recently developed the expertise to send tiny nanodevices made out of DNA directly to the lysosomes of specific immune cells in model organisms such as worms and zebrafish. The two labs teamed up to overcome this challenge.
Kasturi Chakraborty, a former grad student of the Krishnan lab and now a postdoctoral scholar in Becker’s lab, developed a tiny DNA nanodevice that delivered a cysteine protease inhibitor. When Chang Cui, a graduate student in the Becker lab, injected it into a mouse with a tumor, this nanodevice preferentially targeted lysosomes inside TAMs, where it stopped the enzymes from destroying antigens—rendering them once again “visible” to patrolling immune cells.
Combining this DNA nanodevice with frontline chemotherapy led to sustained tumor regression in a triple-negative-breast-cancer model in tests with mice. This outcome was exciting because this type of cancer is particularly difficult to treat.
It’s also a fundamentally different approach from the standard way that researchers think about treating cancer: “When we target a drug, success usually means you have killed the cell you wanted to target,” said Krishnan. “However, in our approach, our intent was not to kill the target cells, but rather reprogram them and change their character. Once the nanodevice flips the switch in a TAM, natural immunity takes care of the rest.”
The researchers hope that this new organelle-specific delivery using DNA nanodevices is the next generation of drug targeting.
It could even go beyond cancer, because specific delivery to macrophages could impact a broad range of diseases where immunity has gone awry, the scientists said.
“You wouldn’t see this work in just a chemistry lab or just an immunology lab,” said Chakraborty. “At UChicago, chemists and biologists are in the same building, so there is an easy flow of interactions and the environment really encourages interdisciplinary science.”
Reference: “A lysosome-targeted DNA nanodevice selectively targets macrophages to attenuate tumours” by Chang Cui, Kasturi Chakraborty, Xu Anna Tang, Kelly Q. Schoenfelt, Alexandria Hoffman, Ariane Blank, Blake McBeth, Natalie Pulliam, Catherine A. Reardon, Swati A. Kulkarni, Tomas Vaisar, Andrea Ballabio, Yamuna Krishnan and Lev Becker, 11 November 2021, Nature Nanotechnology.
DOI: 10.1038/s41565-021-00988-z
Funding: Women’s Board Faculty Research Startup Funds, Ben May Department Startup Funds (L.B.), the University of Chicago Women’s Board (Y.K.)



Be the first to comment on "Using Tiny Nanodevices Made Out of DNA To Treat Cancer"