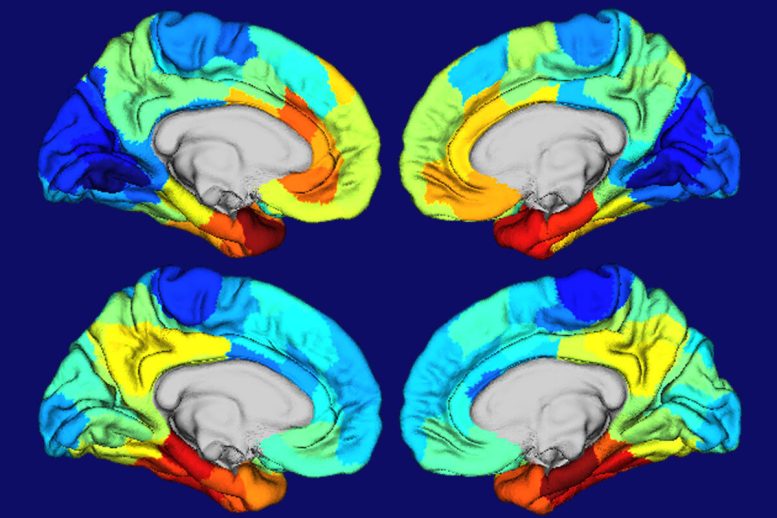
Research findings could help explain rare symptoms such as problems with language and vision.
The first sign of Alzheimer’s disease is typically memory loss, followed by confusion and difficulty thinking. These symptoms reflect the typical pattern of progressively worsening damage to brain tissues. Toxic clusters of proteins first concentrate in the temporal lobes of the brain — the memory area — before spreading to parts of the brain important for thinking and planning.
A new study yields clues as to why certain parts of the brain are particularly vulnerable to Alzheimer’s damage. It comes down to the gene APOE, the greatest genetic risk factor for Alzheimer’s disease. The researchers, from Washington University School of Medicine in St. Louis, found that the parts of the brain where APOE is most active are the areas that sustain the most damage.
Published on November 16 in the journal Science Translational Medicine, the findings help explain why symptoms of Alzheimer’s disease sometimes vary. They also highlight an understudied aspect of Alzheimer’s disease that suggests yet-to-be-discovered biological mechanisms may play an important role in the disease.
“There are some rare, atypical forms of Alzheimer’s in which people first develop language or vision problems rather than memory problems,” said senior author Brian A. Gordon, PhD, an assistant professor of radiology at the School of Medicine’s Mallinckrodt Institute of Radiology.
“When you scan their brains, you see damage to the language or the visual areas, and not so much to the memory areas. People with atypical Alzheimer’s are often screened out of research studies because it’s easier to study a group where everyone has the same set of symptoms. But this heterogeneity tells us that there are things we still don’t understand about how and why Alzheimer’s develops the way it does. There’s a reason why certain brain areas become damaged and not others, and we don’t know that reason yet. Every mystery we uncover with this disease pushes us closer to what we need to address it.”
Protein Accumulation and Brain Atrophy
Alzheimer’s disease begins with a brain protein known as amyloid beta. The protein starts building up into plaques two decades or more before people show the first signs of neurological problems. After years of amyloid accumulation, tangles of tau — another brain protein — begin to form. Soon after, tissues in the affected areas begin to wither and die, and cognitive decline sets in.
To understand why Alzheimer’s brain damage occurs where it does, Gordon and colleagues — including first author Aylin Dincer, a technician in Gordon’s lab — studied 350 people who volunteer for memory and aging studies through the School of Medicine’s Charles F. and Joanne Knight Alzheimer Disease Research Center. The participants underwent brain scans so the researchers could measure the amount and location of amyloid plaques and tau tangles, and the volumes of various brain areas.
Patterns of protein clumps and tissue damage in the volunteers were compared to the gene expression patterns of APOE and other genes associated with Alzheimer’s disease as depicted in the Allen Human Brain Atlas, a detailed map of gene expression in the human brain compiled by the Allen Institute for Brain Sciences.
Genetics and Regional Susceptibility
“There was a close match between where you see high APOE expression, and where you see tau tangles and tissue damage,” said Gordon, also an assistant professor of psychological & brain sciences. “And not just APOE. If you look at, say, the top 20 genes associated with Alzheimer’s disease, they are all expressed in the temporal lobes in similar patterns. There’s something fundamentally different about these regions that make them vulnerable to Alzheimer’s brain damage, and that difference is probably baked in from birth and influenced by a person’s genetics.”
Everyone carries some version of the APOE gene, but people who carry the APOE4 variant are up to 12 times more likely to develop Alzheimer’s disease than the general population, and at a younger age. Alzheimer’s researchers have long known that APOE4 increases the accumulation of amyloid beta in people’s brains. Studying mice that develop tau tangles but not amyloid plaques, David Holtzman, MD, the Barbara Burton and Reuben M. Morriss III Distinguished Professor of Neurology, and colleagues showed that APOE4 also increases damage due to tau, even without amyloid present.
To assess the effect of the high-risk variant of APOE on tau-related brain damage in people, the researchers classified each participant as carrying the high-risk variant or not, and analyzed the protein clusters and atrophy in their brains.
“APOE4 carriers are more likely to start accumulating amyloid, which puts them on the path to Alzheimer’s,” Gordon said. “Then, for the same amount of amyloid they get more tau tangles, which leads to more atrophy. It’s a double hit on the brain.”
Future Research into Gene Expression and Symptoms
In future work, Gordon and colleagues plan to explore how patterns of gene expression relate to patterns of tau damage in people with atypical Alzheimer’s.
“When we see someone who presents with vision problems, is there a specific genetic signature that corresponds to the areas that are damaged in the brain?” Gordon asked. “We want to know why some people have these altered patterns and what it means about how Alzheimer’s disease develops and how it can be treated.”
Reference: “APOE e4 genotype, amyloid-ß, and sex interact to predict tau in regions of high APOE mRNA expression” by Aylin Dincer, Charles D. Chen, Nicole S. McKay, Lauren N. Koenig, Austin McCullough, Shaney Flores, Sarah J. Keefe, Stephanie A. Schultz, Rebecca L. Feldman, Nelly Joseph-Mathurin, Russ C. Hornbeck, Carlos Cruchaga, Suzanne E. Schindler, David M. Holtzman, John C. Morris, Anne M. Fagan, Tammie L.S. Benzinger and Brian A. Gordon, 16 November 2022, Science Translational Medicine.
DOI: 10.1126/scitranslmed.abl7646
Funding: NIH/National Institutes of Health, Alzheimer’s Association, National Science Foundation
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
As presently recalled, earlier studies revealed that more active areas of the brain receive more blood than less active areas. Add to that the more physically sedentary/mentally active lives and the acidic nature of blood in persons with medically undiagnosed long-term chronic food allergy/additive (e.g., added ‘cultured-free’ MSG, minimally) related calcium deficiencies (blood pH optimization with standard serum testing for calcium being unreliable) with inadequate dietary supplies thereof, especially mothers and postmenopausal women not on hormone therapy (estrogen being protective against uric acid), and you have a non-genetic explanation of the causes of the growing incidence/prevalence of dementia and Alzheimer’s Disease. That is, of course, unless the food sensitivities themselves are genetic (some being acquired; some being inherited). As a lay investigator with a family history of food allergies and dementia and two disparate periods of personal diet related short-term memory problems, I postulate the exact biological mechanism is the acidosis eroding cholesterol from the myelin sheaths of brain and other nerve cells with the body trying to repair the damage using whatever else is available (e.g., amyloid beta and tau; plaques and tangles). Perhaps the research should be less ‘treatment’ and more ‘prevention’ oriented.