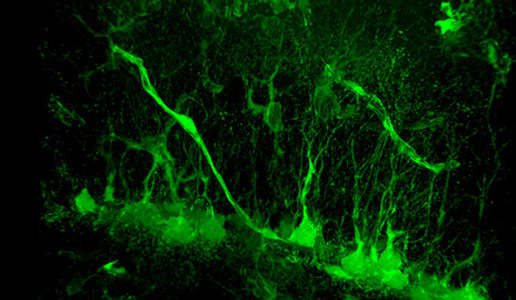
A newly published study from Yale University shows that adult hippocampal neural stem cells (NSCs) express vascular endothelial growth factor receptor (VEGFR) 3 and its ligand VEGF-C, which activates quiescent NSCs to enter the cell cycle and generate progenitor cells.
Scientists have discovered that the human brain can produce new neurons, but exactly how those cells are produced and what purpose they serve are not well understood. Now a study by Yale researchers shows that key developmental factors that control the formation of blood vessels are also necessary for activating brain stem cells.
Yale neurobiologist Jean-Leon Thomas and colleagues, in collaboration with vascular biologist Anne Eichmann, found that neural stem cells lacking a receptor for vascular endothelial growth factor (VEGFR3) produce fewer new brain cells in the hippocampus of mice. In addition, mice lacking VEGFR3 exhibit more anxiety than mice with intact receptors in stem cells.
Surprisingly, said the researchers, a related VEGF molecule does not stimulate brain blood vessels at doses that activate brain stem cells, which suggests that this factor may be used specifically in the treatment of neurological diseases. The team, including first author Jinah Han, reported their findings February 19 in the journal Cell Reports.
Reference: “Vascular Endothelial Growth Factor Receptor 3 Controls Neural Stem Cell Activation in Mice and Humans” by Jinah Han, Charles-Félix Calvo, Tae Hyuk Kang, Kasey L. Baker, June-Hee Park, Carlos Parras, Marine Levittas, Ulrick Birba, Laurence Pibouin-Fragner, Pascal Fragner, Kaya Bilguvar, Ronald S. Duman, Harri Nurmi, Kari Alitalo, Anne C. Eichmann and Jean-Léon Thomas, 19 February 2015, Cell Reports.
DOI: 10.1016/j.celrep.2015.01.049
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.