New discovery reveals that vertebrates gained a unique protein from bacteria over half a billion years ago.
Humans and other backbone-bearing organisms possess a marvel of evolution: eyes that operate similarly to cameras, offering a finely tuned visual system. Charles Darwin acknowledged the eye’s intricacy as a significant potential stumbling block to his theory of natural selection through incremental evolutionary steps.
The distinction in visual capabilities between vertebrates and invertebrates can be traced back to a distinctive protein. This protein is responsible for specializing cells that play a vital role in vision. Mutations in the protein, called IRBP, have been known to cause a variety of diseases such as retinitis pigmentosa, but its evolutionary origin has remained elusive, with no obvious genetic precursor.
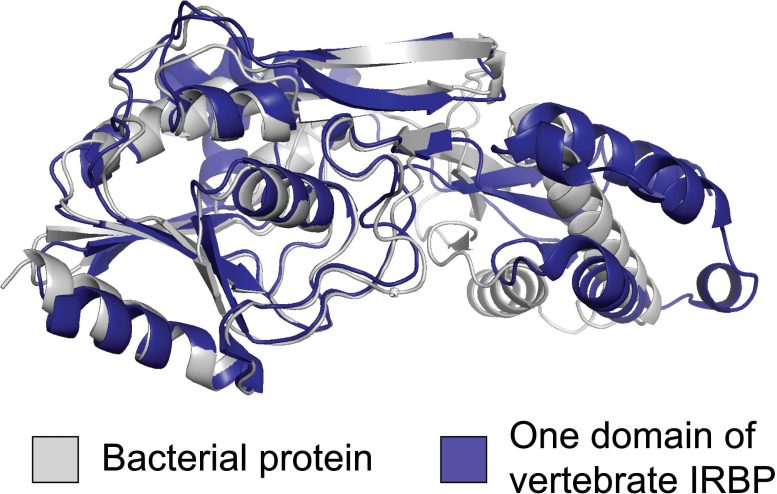
Scientists in the University of California San Diego School of Biological Sciences, publishing in the Proceedings of the National Academy of Sciences, have now traced the 500 million-year-old origin of vertebrate IRBP to a bacterial source. Their discovery, using phylogenetic reconstruction methods, was made possible because of the growing number of fully detailed genomes now available. Their analysis of more than 900 genomes across the tree of life revealed that the IRBP integration in vertebrate eyes was not the result of traditional vertical gene transfer, in which an evolutionary advancement is adapted, or “tinkered with” using available genetic material. Rather, the IRBP was acquired, duplicated, and integrated through horizontal gene transfer from foreign bacterial genes.
“It’s a massive shift because this is an entirely new piece of genetic material that’s been introduced from bacteria,” said Biological Sciences Associate Professor Matt Daugherty, the paper’s senior author. Former UC San Diego undergraduate student Chinmay Kalluraya led the study, and UC San Diego graduate students Alexander Weitzel and Brian Tsu contributed computational expertise. “This study shows that a major innovation that distinguishes vertebrate eyes from all the rest of the eyes out there wasn’t done by molecular tinkering but rather a big leap of genetic innovation.”
How Bacterial Genes Enhanced Vertebrate Vision
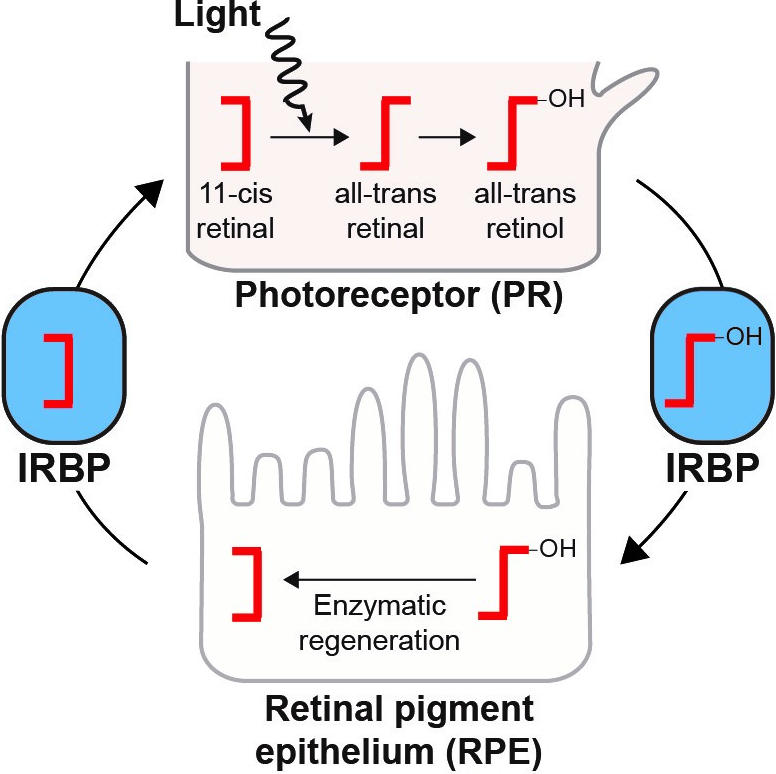
Once the key gene that eventually became IRBP was acquired from bacteria, a new door opened in vertebrates that allowed retinoids, molecules in the eye that directly sense light, to be shuttled between cell types to efficiently recycle it for further light sensing. This separation of photoreception, or light sensing, and retinoid recycling provides unique functionality to vertebrates and the way they can see.
“In order to see in different wavelengths, there needs to be enough light around and that’s one of the arguments for why we can see in the dark really well—we have this enzymatic recycling system that many invertebrates don’t seem to have,” said Daugherty, a researcher in the Department of Molecular Biology. “Eyes are diverse and complicated, and we’ve gone down this path because of this system.”
With more genomes from more organisms becoming available, the researchers believe that other critical functions and systems will similarly trace their roots to bacteria.
“This reshapes the way that we think about evolution and the way we think about complex structures that seem like they’ve emerged out of nowhere,” said Daugherty.
Reference: “Bacterial origin of a key innovation in the evolution of the vertebrate eye” by Chinmay A. Kalluraya, Alexander J. Weitzel, Brian V. Tsu and Matthew D. Daugherty, 10 April 2023, Proceedings of the National Academy of Sciences.
DOI: 10.1073/pnas.2214815120
The study was funded by the National Institutes of Health, Pew Biomedical Scholars, the Burroughs Wellcome Fund Investigators in the Pathogenesis of Infectious Disease, UC San Diego’s Halıcıoğlu Data Science Institute, UC San Diego Triton Research, and Experimental Learning Scholars.
In addition to Kalluraya, a Selma and Robert Silagi Award for Undergraduate Excellence winner at UC San Diego and now a graduate student at MIT, Weitzel, Tsu, and Daugherty coauthored the paper.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.