Clinical studies reveal extended survival in diffuse midline glioma patients receiving ONC201 treatment; research also explains the underlying mechanism of the drug’s success.
For the first time, a potential drug candidate has been identified by researchers that show promise in improving outcomes for patients suffering from a specific type of childhood brain tumor that currently lacks effective treatment options. The compound, named ONC201, was observed to nearly double the survival rates for patients diagnosed with diffuse midline glioma (DMG) or diffuse intrinsic pontine glioma (DIPG) when compared to prior patient outcomes.
The findings are reported by an international team of researchers led by the University of Michigan Health Rogel Cancer Center and the Chad Carr Pediatric Brain Tumor Center.
In addition to reporting on the results of two early-stage clinical trials, the paper reveals the underlying mechanisms behind the compound’s success in these tumors. The paper is published in Cancer Discovery, a journal of the American Association for Cancer Research.
The Deadly Challenge of H3K27M-Mutated Tumors
Diffuse midline gliomas including DIPG with a mutation called H3K27M are particularly aggressive, with an overall survival rate of 11-15 months. These tumors are most frequently found in children and young adults. The only available treatment is radiation, and even that is difficult as the tumors are located amid brain regions with critical functions.
“It’s an incredibly difficult tumor to treat,” said senior author Carl Koschmann, M.D., associate professor of pediatric neuro-oncology and clinical scientific director of the Chad Carr Pediatric Brain Tumor Center at Michigan Medicine. “Prior to this study, there have been more than 250 clinical trials that have not been able to improve outcomes. This is a major crack in the armor.”
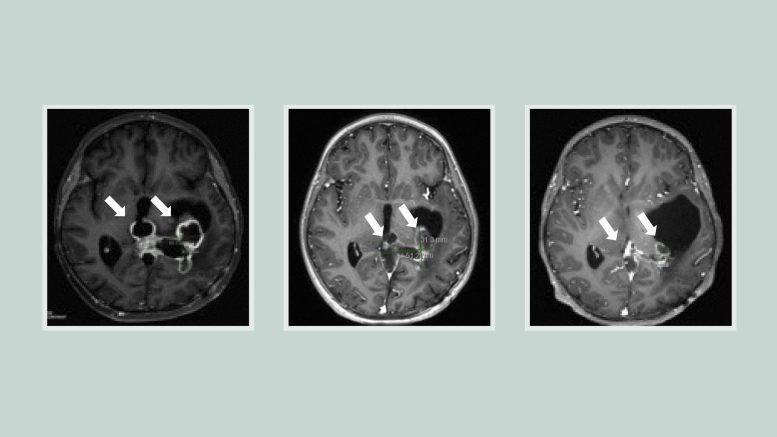
In two clinical trials testing ONC201 in a total of 71 patients with H3K27M-mutated diffuse midline gliomas, the median overall survival was nearly 22 months for tumors that had not recurred at the time of enrollment. Almost a third of the patients lived longer than two years.
ONC201 took an unusual path to a clinical trial. Initially designed to target dopamine receptors, which are upregulated in many different tumors, researchers saw that the drug passes the blood-brain barrier, one of the biggest challenges to designing drugs for brain tumors. Initial trials in glioblastoma were not successful, but a small number of patients with DMG that carried the H3K27M mutation had more promising results. Without understanding why it worked better in these patients, a phase 1 trial was started in children and young adults with H3K27M-mutated DMG.
Meanwhile, Koschmann and co-author Sriram Venneti, M.D., Ph.D., were trying to figure out what was happening in these tumor cells.
Metabolic and Epigenetic Effects of ONC201
Through the trial, they collected cerebrospinal fluid from patients. They used this fluid to analyze metabolic changes and found ONC201 got into the tumor cells and affected mitochondria. Patients who responded to the drug had an increase in a metabolite called L-2HG produced by tumor cells.
Koschmann called the finding “very much unexpected.” The team found that increased L-2HG reversed tumor-defining epigenetic signals causing tumor cells to differentiate more and divide less. The longer patients were on ONC201, the more tumors exhibited these epigenetic reversals.
“This could explain why this patient population was responding so well to the drug because it had this specific epigenetic abnormality that could be turned off by ONC201. The tumors have an epigenetic change caused by the H3K27M mutation and ONC201 metabolically undoes that change,” said Venneti, associate professor of pathology and pediatrics and scientific research director of the Chad Carr Pediatric Brain Tumor Center at Michigan Medicine.
Additional clinical trials are currently underway, including testing ONC201 in combination with other therapies. Researchers at U-M’s Chad Carr Pediatric Brain Tumor Center are also continuing to look at ways to overcome resistance to ONC201 by using drug combinations.
Koschmann notes that even a near-doubling of survival is not enough for families of patients with this diagnosis, as the tumor remains very lethal. But he hopes this first step will lead to bigger leaps in the future.
“For now we have this patient population that didn’t have a drug before, and now we see many of the tumors responding. We have a platform to build on and we can also explain why it’s working,” he said.
“We are really excited about this study and envision ONC201 becoming the standard of care for these patients in the near future,” Venneti said.
Reference: “Clinical efficacy of ONC201 in H3K27M-mutant diffuse midline gliomas is driven by disruption of integrated metabolic and epigenetic pathways” by Sriram Venneti, Abed Rahman Kawakibi, Sunjong Ji, Sebastian M. Waszak, Stefan R. Sweha, Mateus Mota, Matthew Pun, Akash Deogharkar, Chan Chung, Rohinton S. Tarapore, Samuel Ramage, Andrew Chi, Patrick Y. Wen, Isabel Arrillaga-Romany, Tracy T. Batchelor, Nicholas A. Butowski, Ashley Sumrall, Nicole Shonka, Rebecca A. Harrison, John de Groot, Minesh Mehta, Matthew D. Hall, Doured Daghistani, Timothy F. Cloughesy, Benjamin M. Ellingson, Kevin Beccaria, Pascale Varlet, Michelle M. Kim, Yoshie Umemura, Hugh Garton, Andrea Franson, Jonathan Schwartz, Rajan Jain, Maureen Kachman, Heidi Baum, Charles F. Burant, Sophie L. Mottl, Rodrigo T. Cartaxo, Vishal John, Dana Messinger, Tingting Qin, Erik Peterson, Peter Sajjakulnukit, Karthik Ravi, Alyssa Waugh, Dustin Walling, Yujie Ding, Ziyun Xia, Anna Schwendeman, Debra Hawes, Fusheng Yang, Alexander R. Judkins, Daniel Wahl, Costas A. Lyssiotis, Daniel de la Nava, Marta M. Alonso, Augustine Eze, Jasper Spitzer, Susanne V. Schmidt, Ryan J. Duchatel, Matthew D. Dun, Jason E. Cain, Li Jiang, Sylwia A. Stopka, Gerard Baquer, Michael S. Regan, Mariella G. Filbin, Nathalie Y.R. Agar, Lili Zhao, Chandan Kumar-Sinha, Rajen Mody, Arul Chinnaiyan, Ryo Kurokawa, Drew Pratt, Viveka N. Yadav, Jacques Grill, Cassie Kline, Sabine Mueller, Adam Resnick, Javad Nazarian, Joshua E. Allen, Yazmin Odia, Sharon L. Gardner and Carl Koschmann, 16 August 2023, Cancer Discovery.
DOI: 10.1158/2159-8290.CD-23-0131
Funding for this work is from the National Institutes of Health, Department of Defense, University of Michigan Chad Carr Pediatric Brain Tumor Center, The Evans Family, ChadTough Defeat DIPG Foundation, Catching Up with Jack, Pediatric Brain Tumor Foundation, Prayers From Maria Foundation, Michael Miller Memorial Foundation, Morgan Behen Golf Classic, Yuvaan Tiwari Foundation, Sontag Foundation, Alex Lemonade Stand Foundation, Hyundai Hope Foundation, Research Council of Norway, The South-Eastern Norway Regional Health Authority, The University of Oslo, Oncoceutics, Inc., Chimerix, Inc., Making Headway Foundation, Gustave Roussy Foundation Pediatric Campaign, Imagine for Margo Charity, National Research Foundation of Korea, DGIST Start-up Fund, and University of Michigan Taubman Institute.
This work was supported by these Rogel Cancer Center Shared Resources: Experimental Irradiation and Preclinical Molecular Imaging
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.