For the first time, scientists at the Morgridge Institute for Research have generated near-atomic resolution images of a major viral protein complex responsible for replicating the RNA genome of a member of the positive-strand RNA viruses, the large class of viruses that includes coronaviruses and many other pathogens.
The results should aid development of new types of antivirals and provide mechanistic insights into the virus life cycle.
“The rapidly advancing ability to visualize such crucial structures is game-changing,” says Paul Ahlquist, director of the John W. and Jeanne M. Rowe Center for Virology Research at the Morgridge Institute and professor of oncology and molecular virology at the University of Wisconsin-Madison. Other authors of the study included Nuruddin Unchwaniwala, Hong Zhan, Janice Pennington, Mark Horswill, and Johan den Boon.
Using an advanced technique called cryoelectron microscope (cryo-EM) tomography, Ahlquist and his team built upon their previous work, which first revealed the existence of this crown-like viral RNA replication complex.
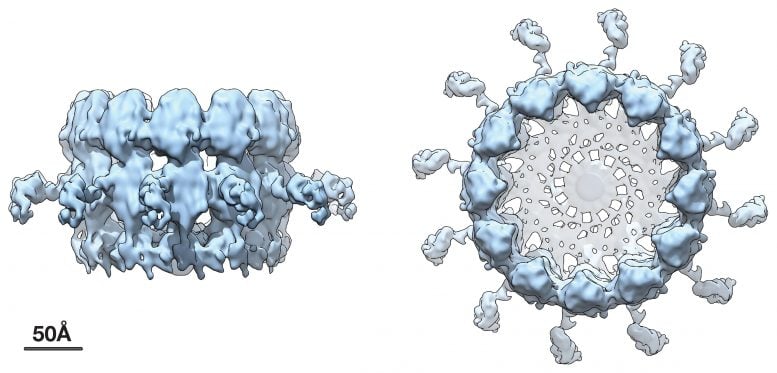
The new research, published July 20 in the Proceedings of the National Academy of Sciences (PNAS), shows the replication crown complex at a dramatically improved resolution of approximately 8.5 angstroms, which corresponds to the spacing of a few atoms.
“Cryo-EM has recently gone through a quantum leap in its capabilities,” Ahlquist says. “In this study our research group combined multiple advances to greatly improve sample preparation, image acquisition, and image processing, and to map the position of specific protein domains in the complex.”
Advanced Cryo-EM imaging shows an approximate fourfold increase in resolution from prior imaging, revealing the three major subdomains that make up the viral RNA replication crown complex.
The positive-strand RNA viruses addressed in this work are the largest of six genetic classes of viruses and include many important pathogens such as the Zika, dengue, and chikungunya viruses, as well as coronaviruses like SARS-CoV-2, cause of the current COVID-19 pandemic.
In each positive-strand RNA virus, most of the viral genes are devoted to a single process: replicating the viral RNA genome.
“Given this massive investment of resources, viral RNA genome replication is arguably one of the most important processes in infection, and It is already a major target for virus control,” Ahlquist says.
Within an infected cell, viral RNA replication occurs at modified cellular membranes, often in association with spherules, virus-induced vesicles approximately 50–100 nanometers in size. Ahlquist and his team previously showed that in each such genome replication complex, a copy of the viral RNA genome or chromosome is protected inside the spherule vesicle to function as a replication template. The replication complex repeatedly copies this archival viral RNA chromosome to produce new progeny genomes that are released through a membranous neck on the vesicle into the cytoplasm, where they are incorporated as the payload of new infectious virions.
This prior work further showed that the key viral protein that induces the replication of vesicles and copies the viral RNA resides in a striking ring or crown structure that sits atop the cytoplasmic side of the spherule neck that connects with the cytoplasm.
The new higher resolution cryo-EM images and complementary results show that the crown is composed of twelve copies of the key viral RNA replication protein arranged like staves in a barrel. Additionally, the images revealed zipper-like interactions that act like hoops on a barrel to join adjacent segments together to form the ring-like crown. These zippering interactions correspond well with multimerizing interactions that the Ahlquist group has previously mapped in this protein.
The viral RNA replication protein that forms the crown is an extremely large, multi-domain, multi-functional protein, nearly 1000 amino acids in size. This protein contains RNA polymerase and RNA capping domains— two enzymatic domains that are conserved across numerous positive-strand RNA viruses for synthesizing new viral genome copies—plus other domains for multimerizing, binding membranes, and other functions.
How these domains are physically organized in the crown structure is one of the most important issues for understanding how the replication complex functions, and was one of several strong motivations for defining the high-resolution crown structure.
Using an approach that combined a genetically engineered, site-specific tag with labeling by nanoscale gold particles visible in cryo-EM, the researchers found that the C-terminal polymerase end of the viral RNA replication protein is positioned at the apex of the crown, leaving the N-terminal capping domain at the bottom of the structure to interact with the membrane.
This apical position of the polymerase has important mechanistic implications for early steps in the replication process that recruit the starting viral RNA template into the complex and form the replication vesicle, as well as for later steps in which the template is copied to make new progeny genomes to be packaged into infectious virus particles. These results provide a strong foundation for further experiments to define the replication complex structure and function at even higher levels.
“We hope to continue to improve the RNA replication complex crown structure to provide additional important refinements in future,” Ahlquist says. “We also hope to address growing indications from our work that conformational changes in these proteins are critical to their multiple functions.”
“Such advances will reveal in increasing detail how these complexes assemble and operate, and thus how they might be best attacked,” he adds. “These insights should provide the basis for novel, stronger antiviral mechanisms.”
Reference: “Subdomain cryo-EM structure of nodaviral replication protein A crown complex provides mechanistic insights into RNA genome replication” by Nuruddin Unchwaniwala, Hong Zhan, Janice Pennington, Mark Horswill, Johan A. den Boon and Paul Ahlquist, 20 July 2020, Proceedings of the National Academy of Sciences.
DOI: 10.1073/pnas.2006165117
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.