A new study published in the Journal of Alzheimer’s Disease (JAD) on January 13, 2020, supports a growing body of research that links human exposure to aluminum with Alzheimer’s disease (AD). Researchers found significant amounts of aluminum content in brain tissue from donors with familial AD. The study also found a high degree of co-location with the amyloid-beta protein, which leads to the early onset of the disease.
“This is the second study confirming significantly high brain accumulation in familial Alzheimer’s disease, but it is the first to demonstrate an unequivocal association between the location of aluminum and amyloid-beta in the disease. It shows that aluminum and amyloid-beta are intimately woven in the neuropathology,” explained lead investigator Christopher Exley, Ph.D., Birchall Center, Lennard-Jones Laboratories, Keele University, Staffordshire, UK.
“[T]he new research confirms my resolve that within the normal lifespan of humans, there would not be any AD if there were no aluminum in the brain tissue. No aluminum, no AD.” Dr. Christopher Exley
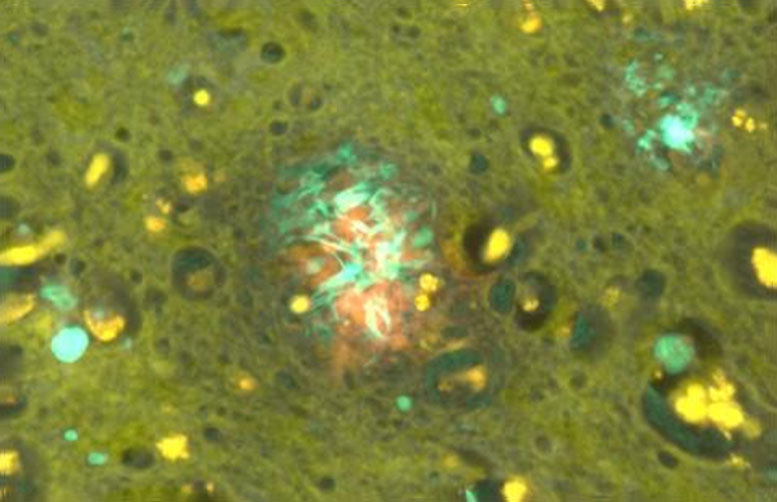
An association between aluminum and amyloid-beta has been suggested for over 40 years. In an earlier study, brain tissue from donors in the United Kingdom diagnosed with familial AD showed significant accumulations of aluminum. To further understand this relationship, in the current study the researchers measured aluminum in the brain tissue of a cohort of Colombian donors with familial AD who shared a specific mutation. The mutation leads to elevated levels of amyloid-beta, early disease onset, and an aggressive disease etiology. The levels were compared with a control set of brain tissues from donors with no diagnosis of neuropathological disease. They also used aluminum-specific fluorescence microscopy imaging to investigate the relationship between aluminum and amyloid-beta in familial AD.
The results were striking. The aluminum content of the brain tissue from donors with the genetic mutation was universally high, with 42% of tissues having a level considered pathologically significant, and the levels were significantly higher than those in the control set. The imaging studies identified aluminum deposits in all brain tissues studied. They were predominantly co-located with amyloid-beta in senile plaques and occasionally in the brain vasculature. Aluminum was also found separately from amyloid-beta in intracellular compartments including glia and neuronal axons. The results strongly suggest that genetic predispositions known to increase amyloid-beta in brain tissue also predispose individuals to accumulate and retain aluminum in brain tissue.
“Compelling localization of aluminum with a central player in AD, amyloid-beta, strengthens the link of aluminum to the pathogenesis of AD,” commented George Perry, Ph.D., Professor of Biology, Semmes Distinguished University Chair in Neurobiology, University of Texas at San Antonio, and Editor-in-Chief of JAD.
“One could envisage increased amyloid-beta in brain tissue as a response to high levels of aluminum content, or that aluminum fosters the accumulation of amyloid-beta,” said Dr. Exley. “Either way, the new research confirms my resolve that within the normal lifespan of humans, there would not be any AD if there were no aluminum in the brain tissue. No aluminum, no AD.”
Reference: “Aluminum and Amyloid-β in Familial Alzheimer’s Disease” by Matthew Mold, Caroline Linhart, Johana Gómez-Ramírez, Andrés Villegas-Lanau and Christopher Exley, 13 January 2020, Journal of Alzheimer’s Disease.
DOI: 10.3233/JAD-191140
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
18 Comments
what type of everyday products are we talking about with aluminum
Is betty crocker foil wrap one of them
Largely irrelevant. Aluminium in normal household wares won’t have any effect in anyone with healthy kidney function.
Old-style jams, made in aluminium cookware, were chock full of aluminium. Perfectly safe unless kidney function was compromised. Even then, you had to consume a lot.
The author of this study is a known antivaxxer who has dedicated his entire career to trying to prove aluminum adjuvants in vaccines cause alzheimer’s. His mentor, Exley, has similarly attempted to use manipulated data and embarrassingly flawed studies to prove aluminum in vaccines causes autism. As well, the studies they base they’re current research on were not only flawed, but thoroughly debunked. SciTech needs to do a much better job vetting the material they use in their articles.
Thank you for sharing
Thanks for that. The quote: “Either way, the new research confirms my resolve….” His “resolve” is nothing but a belief, and it suggests to me that he has an agenda. I have no idea how accurate his conclusions are, but it’s a bad sign when a scientist openly displays beliefs and biases. It seriously degrades the requirement of objectivity that is a requirement for all good science.
You REALLY should not talk out your rear-end.
You clearly have no critical thinking abilities, as well as doing some real research on your own. Stop regurgitating what you hear on the nightly news—-which is wholly-owned by the pharmaceutical industry.
At least get the headline right: Aluminum accumulation (not ‘exposure’) linked to
Alzheimer’s.
That would be scientific precision.
Observed by the University of Manchester, in collaboration with SERC Daresbury Laboratory, in the 1990s.
Has anyone looked into high levels of Aluminum oxide in deoderants and anti perspirants.
Yeah, this guy is a notorious crank and he’s using an aluminum stain at a concentration 3 ORDERS OF MAGNITUDE higher than necessary and is missing crucial controls.
Bad, agenda-driven science is easier than real science. The media and readers should exercise scientific skepticism, especially about hyped and sensational claims like this, supported by a single, poor-quality, small, study published in a low-impact journal. Next time call up one of the zillions of people who call Exley a wingnut and ask *them* to explain the flaws in the study instead of regurgitating it without a thought.
Why are so many people showing outrage that aluminium ‘acummulation’ could be harmful you should encourage more research before deciding its not feasible
Do you guys work in the aluminum trade or something?
Considering Dr. Exley has been studying aluminium for over 30 years, I would think he knows more than most on the subject. I would like to be educated on what types of aluminium we should be avoiding. I already avoid vaccines since it has been proven throughout history to cause neurologial disorders especially when injected.
Aluminum is a toxin. It’s sad so many fail to grasp that, lashing out at ‘anti-vaxxers’ as ignorant, evil, and the reason diseases spread. Big Pharma has duped many with their propaganda. God gave you a immune system that works as it’s designed to work. Proper nutrition and hygiene will get you through illnesses better than vaccines that bring on the illnesses society suffers from. Wonder who’s buttering their bread? hmmm…..
Pharmaceutical companies rely upon ill health in the population to survive and reap their profits. No drug company has a vested interest in curing disease. They do, however, have a massive vested interest in maintaining ill-health, creating disease and manufacturing chemicals which will promote this under the guise of ‘therapy’ for the symptoms – rarely ever the cause – of disease. Dr John Braithwaite, now a Trade Practices Commissioner, in his expose, Corporate Crime in the Pharmaceutical Industry, states:
Wow very nicely said!
The glymphatic network deals with waste clearance in the brain.
As it was only just discovered a few years ago I do wonder how the century-long use of vaccines has been properly studied with respect to scattering aluminum adjuvants like buckshot throughout this system in the brain . . .
During a three hour period the Manila VA ordered 3 different nurses to inject with Aluminum Loaded Adjuvants. The VA claimed the injections were for the Flu, Pneumonia, and another illness! I am now suffering DEMENTIA and my life has been a living hell! ALUMINUM DOES INDEED CAUSE DEMENTIA AND MUCH MORE!