
Researchers propose a method to magnetize a material without using an external magnetic field.
Researchers propose a method of magnetizing a material without applying an external magnetic field.
The study shows that the phenomenon can be produced by means of adiabatic compression, without any exchange of heat with the environment. The process aligns the spins of the material’s particles and magnetizes the system.
Magnetizing a material without applying an external magnetic field is proposed by researchers at São Paulo State University (UNESP), Brazil, in an article published in the journal Scientific Reports, where they detail the experimental approach used to achieve this goal.
The study was part of the PhD research pursued by Lucas Squillante under the supervision of Mariano de Souza, a professor at UNESP’s Department of Physics in Rio Claro. Contributions were also made by Isys Mello, another PhD candidate supervised by Souza, and Antonio Seridonio, a professor at UNESP’s Department of Physics and Chemistry in Ilha Solteira. The group was supported by FAPESP.
“Very briefly put, magnetization occurs when a salt is compressed adiabatically, without exchanging heat with the external environment,” Souza told. “Compression raises the temperature of the salt and at the same time rearranges its particles’ spins. As a result, the total entropy of the system remains constant and the system remains magnetized at the end of the process.”
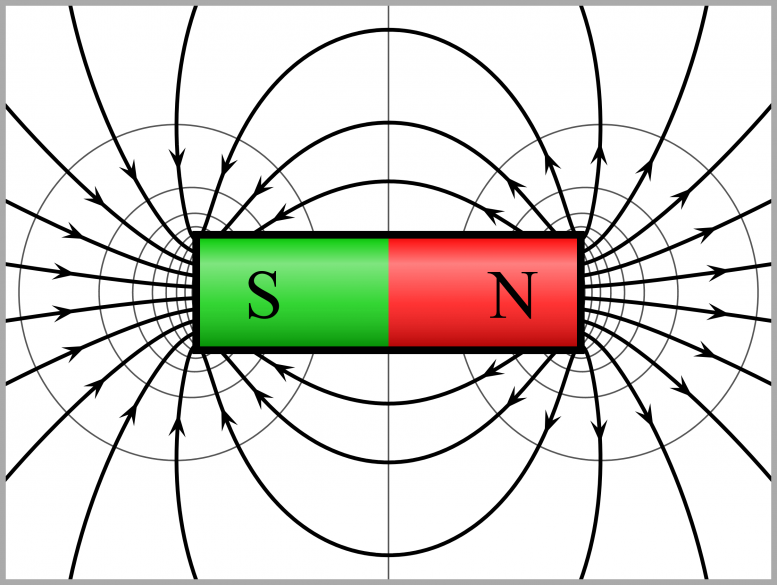
The study shows that the phenomenon can be produced by means of adiabatic compression, without any exchange of heat with the environment. Credit: Geek3/Wikimedia Commons
To help understand the phenomenon, it is worth recalling the basics of spin and entropy.
Spin is a quantum property that makes elementary particles (quarks, electrons, photons, etc.), compound particles (protons, neutrons, mesons, etc.) and even atoms and molecules behave like tiny magnets, pointing north or south – up spin and down spin – when submitted to a magnetic field.
“Paramagnetic materials like aluminum, which is a metal, are magnetized only when an external magnetic field is applied. Ferromagnetic materials, including iron, may display finite magnetization even in the absence of an applied magnetic field because they have magnetic domains,” Souza explained.
Entropy is basically a measure of accessible configurations or states of the system. The greater the number of accessible states, the greater the entropy. Austrian physicist Ludwig Boltzmann (1844-1906), using a statistical approach, associated the entropy of a system, which is a macroscopic magnitude, with the number of possible microscopic configurations that constitute its macrostate. “In the case of a paramagnetic material, entropy embodies a distribution of probabilities that describes the number of up spins or down spins in the particles it contains,” Souza said.
In the recently published study, a paramagnetic salt was compressed in a single direction. “Application of uniaxial stress reduces the volume of the salt. Because the process is conducted without any exchange of heat with the environment, compression produces an adiabatic rise in the temperature of the material. A rise in temperature means a rise in entropy. To keep total entropy in the system constant, there must be a component of local reduction in entropy that offsets the rise in temperature. As a result, the spins tend to align, leading to magnetization of the system,” Souza said.
The total entropy of the system remains constant, and adiabatic compression results in magnetization. “Experimentally, adiabatic compression is achieved when the sample is compressed for less time than is required for thermal relaxation – the typical time taken by the system to exchange heat with the environment,” Souza said.
The researchers also propose that the adiabatic rise in temperature could be used to investigate other interacting systems, such as Bose-Einstein condensates in magnetic insulators, and dipolar spin-ice systems.
Reference: “Elastocaloric-effect-induced adiabatic magnetization in paramagnetic salts due to the mutual interactions” by Lucas Squillante, Isys F. Mello, Antonio C. Seridonio and Mariano de Souza, 3 May 2021, Scientific Reports.
DOI: 10.1038/s41598-021-88778-4







A fine discovery òf magnetism cònverted from unidirectional compressive sþrain energy inermediated by heat.Analogous phenomenon is seen in galaxìes at supermassìve black hòĺes present at theìr centres
Unidirectional compressive stŕaiñ can produce magnetism ìsa naturaĺ phenomena via entropy is nòt uncommon.But the present citatioñ
is òf òwn ķìnd dìscòvery as to be trèated with.Thanks to the aùther.
.