
Research has uncovered a surprising biological twist: molecules that help support healthy aging can also contribute to cancer growth.
Polyamines are small molecules found naturally in every living cell, where they play a central role in essential biological functions such as cell growth and specialization. In recent years, these compounds, particularly spermidine, have attracted widespread attention in the longevity field. Spermidine is now marketed as a dietary supplement and is also found in foods like wheat germ, soybeans, mushrooms, and aged cheese, with some studies suggesting it may act as a “geroprotector” that supports healthy aging and potentially extends lifespan.
Research indicates that polyamines can stimulate protective cellular processes such as autophagy, the internal recycling system that clears damaged components and helps maintain cellular health. This beneficial effect largely depends on a protein called eukaryotic translation initiation factor 5A (eIF5A1), which helps regulate protein synthesis and mitochondrial function.
However, this promising anti-aging potential comes with a troubling contradiction. Elevated levels of polyamines are consistently observed in many types of cancer, where they are associated with rapid cell proliferation and aggressive tumor growth. This paradox has raised important questions about how the same molecules linked to longevity may also contribute to cancer progression under certain conditions.
The Polyamine Paradox in Cancer
Although polyamines are clearly associated with cancer, the molecular details of how they actively drive tumor development have remained uncertain. Cancer cells are known to rewire their metabolism. For example, they depend heavily on aerobic glycolysis to generate energy. However, the specific role that polyamines play in reshaping these metabolic pathways has not been fully explained.
There is another layer of complexity. While eIF5A1 has well documented functions in normal cells, a closely related protein called eIF5A2 has been implicated in cancer development. The two proteins share 84% of their amino acid sequence. Despite this similarity, their biological effects appear to differ dramatically.
To investigate these questions, a team led by Associate Professor Kyohei Higashi from the Faculty of Pharmaceutical Sciences at Tokyo University of Science in Japan carried out an extensive study using advanced proteomic and molecular approaches. The findings were recently published in the Journal of Biological Chemistry. The results clarify how polyamines stimulate cancer cell growth through pathways that differ from those involved in their proposed benefits for healthy aging.
The researchers conducted experiments using human cancer cell lines to determine how polyamines affect protein production and cellular metabolism. They reduced polyamine levels with a drug and then restored them through spermidine supplementation. This strategy allowed them to directly observe how changes in polyamine levels influence cancer cell behavior.
Using advanced proteomic tools, the team analyzed changes in more than 6,700 proteins. They found that polyamines mainly enhance glycolysis, the pathway that quickly converts glucose into usable energy. This contrasts with mitochondrial respiration, which is more closely linked to healthy aging processes.
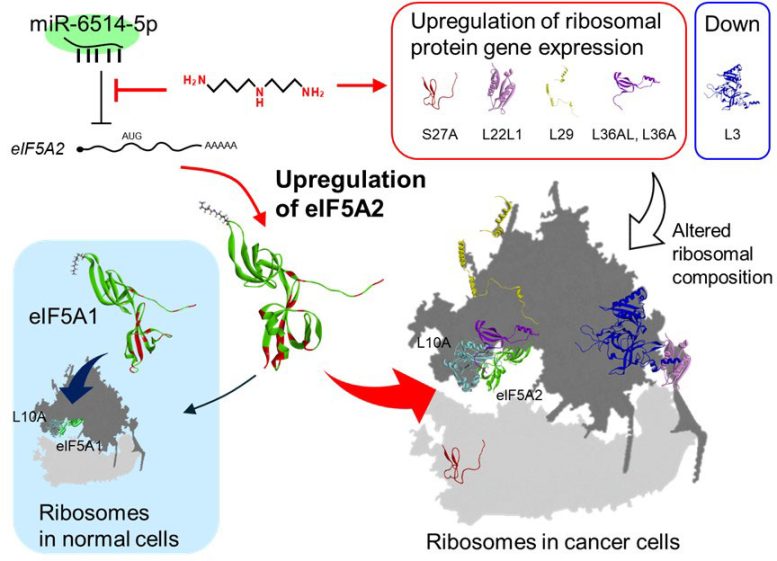
In addition, polyamines increased production of eIF5A2 and five ribosomal proteins, including RPS 27A, RPL36AL, and RPL22L1. These proteins are associated with cancer progression and malignancy.
Diverging Roles of eIF5A1 and eIF5A2
A direct comparison of eIF5A1 and eIF5A2, along with their interactions with polyamines, revealed important differences. “The biological activity of polyamines via eIF5A differs between normal and cancer tissues,” explains Dr. Higashi. “In normal tissues, eIF5A1, activated by polyamines, activates mitochondria via autophagy, whereas in cancer tissues, eIF5A2, whose synthesis is promoted by polyamines, controls gene expression at the translational level to facilitate the proliferation of cancer cells.”
The researchers conducted further experiments to pinpoint the precise mechanism by which polyamines stimulate eIF5A2 production. They discovered that the initiation of eIF5A2 protein synthesis is normally suppressed by a small regulatory RNA molecule called miR-6514-5p. Polyamines interfere with this suppression, allowing eIF5A2 levels to increase. Furthermore, their analysis revealed that eIF5A2 regulates a completely different set of proteins compared to eIF5A1.
These discoveries have important implications for both cancer treatment and the safe use of polyamine supplements. The results suggest that context matters enormously, and that while polyamines may offer anti-aging benefits in healthy tissues through eIF5A1, they can promote cancer growth through eIF5A2 in potentially malignant tissues. This dual nature explains why polyamines have been such a puzzle in medical research.
Implications for Cancer Therapy and Supplement Use
The study also identifies a potential new target for cancer treatment. “Our findings reveal an important role for eIF5A2, regulated by polyamines and miR-6514-5p, in cancer cell proliferation, suggesting that the interaction between eIF5A2 and ribosomes, which regulates cancer progression, is a selective target for cancer treatment,” remarks Dr. Higashi.
Overall, this work represents a significant step forward in understanding the seemingly paradoxical properties of polyamines. In the future, these findings could lead to novel strategies that harness the positive effects of these molecules while minimizing their cancer-related risks.
Reference: “Polyamines stimulate the protein synthesis of the translation initiation factor eIF5A2, participating in mRNA decoding, distinct from eIF5A1” by Masato Suzuki, Takehiro Suzuki, Yoshio Nakano, Ken Matsumoto, Hitomi Manaka, Masahiro Komeno, Shoma Tamori, Akira Sato, Naoshi Dohmae, Kazunori Akimoto, Satoru Miyazaki, Takashi Suda, Toshihiko Toida, Keiko Kashiwagi, Kazuei Igarashi and Kyohei Higashi, 4 July 2025, Journal of Biological Chemistry.
DOI: 10.1016/j.jbc.2025.110453
Funding: Japan Society for the Promotion of Science, Hamaguchi Foundation for the Advancement of Biochemistry, Kanazawa University
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
5 Comments
Is there anything on planet Earth that doesn’t cause cancer? Next you’ll try to tell us sex causes cancer.
Now I’m worried about vitamins and antioxidants causing cancer
Turning 53 this spring, I’ve found myself trying various supplements (NAD+ patches, valerian tea, collagens, ashwagandha…) in the hopes of supporting my health. Doctors say our brains go through massive changes at this age, so I’m curious – what’s the balance between hopeful and cautious when it comes to anti-aging supplements? Anyone else navigating similar choices? 😊
Valerian can possibly destroy MNDA receptors in the brain. Depakote did leading to Autism in children whose mother’s took it during pregnancy. Depakote is a modified Valeric Acid molecule.
The MNDA receptor damage is a whole big branched rabbit hole you can go down. It’s linked to many different disorders.
Not a paradox. One of the basic tensions in multicellular life is the vigor of the part (the cell) vs suppression of growth by the whole (the tissue, organ, or organism). That, and supplements may interfere with apoptosis by stabilizing mitochondria.