Researchers at NIMTE have turned metal corrosion into a tool for efficient biomass upgrading, achieving high HMF-to-BHMF conversion rates with a CoCuMW/CF electrode. Their findings offer a low-cost, sustainable solution for bio-based chemical production.
A research team led by Prof. Jian Zhang from the Ningbo Institute of Materials Technology and Engineering (NIMTE) of the Chinese Academy of Sciences (CAS) has harnessed metal corrosion to develop high-performance electrodes, facilitating the efficient and cost-effective upgrading of bio-based 5-hydroxymethylfurfural (HMF). Their findings were published in Chem Catalysis.
While corrosion is typically associated with material degradation and economic loss, researchers are now investigating its potential for advantageous applications, particularly in biomass upgrading.
As one of the most abundant renewable resources on Earth, biomass can be catalytically converted into fuels and chemicals that replace traditional fossil resources, contributing significantly to the global goal of “peak carbon dioxide emission and carbon neutrality.”
A Novel Approach: Corrosion-Assisted Electrode Fabrication
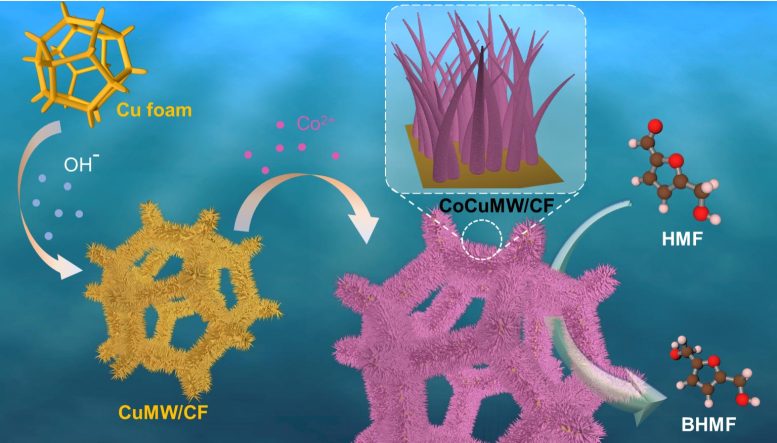
Inspired by the idea of “turning damage into benefits,” the research team combined spontaneous metal corrosion with efficient biomass upgrading. They fabricated CoCu microwire arrays on copper foam (CoCuMW/CF) using cobalt ion-enhanced corrosion induction.
The CoCuMW/CF electrode enables efficient electrochemical reduction of HMF to 2,5-bis(hydroxymethyl)furan (BHMF), which can be further converted into environmentally friendly plastic or rubber products, high-value derivatives, and high-quality bio-based chemicals through simple methods.
Moreover, the prepared CoCuMW/CF electrode demonstrated a remarkable HMF conversion rate of 95.7% and a BHMF yield of 85.4% at the potential of -0.5 V vs. reversible hydrogen electrode (RHE), indicating superior performance for HMF hydrogenation in a neutral electrolyte.
Notably, the activation energy for the HMF electrocatalytic reduction was 16.6 ± 2.5 kJ·mol-1, significantly lower than in thermocatalysis.
Enhanced Catalysis Through Density Functional Theory (DFT) Calculations
In this study, density functional theory (DFT) calculations revealed that the CoCuMW/CF electrode exhibits reduced free energy barriers for both the initial and subsequent hydrogenation steps of HMF. This reduction in energy barriers enhances the catalytic performance and the selectivity for BHMF production.
The study challenges conventional views on corrosion phenomena and enables highly efficient electrochemical hydrogenation using copper-based electrocatalysts for biomass upgrading at extremely low cost. This advancement holds great promise for accelerating HMF electrohydrogenation applications.
Reference: “Corrosion-induced CoCu microwire arrays for efficient electroreduction of 5-hydroxymethylfurfural” by Bin Zhu, Jie Yang, Qiuge Wang, Xiao Yu, Shilin Fan, Weiping Xie, Jian Zhang and Chunlin Chen, 10 February 2025, Chem Catalysis.
DOI: 10.1016/j.checat.2024.101259
This work was supported by the National Natural Science Foundation of China, the Key Research and Development Program of Zhejiang Province, and the Ningbo Science and Technology Bureau, among others.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.