Salk researchers and collaborators show how the protein damages cells, confirming COVID-19 as a primarily vascular disease.
Scientists have known for a while that SARS-CoV-2’s distinctive “spike” proteins help the virus infect its host by latching on to healthy cells. Now, a major new study shows that they also play a key role in the disease itself.
The paper, published on April 30, 2021, in Circulation Research, also shows conclusively that COVID-19 is a vascular disease, demonstrating exactly how the SARS-CoV-2 virus damages and attacks the vascular system on a cellular level. The findings help explain COVID-19’s wide variety of seemingly unconnected complications, and could open the door for new research into more effective therapies.
Unifying COVID Complications Through Vascular Insight
“A lot of people think of it as a respiratory disease, but it’s really a vascular disease,” says Assistant Research Professor Uri Manor, who is co-senior author of the study. “That could explain why some people have strokes, and why some people have issues in other parts of the body. The commonality between them is that they all have vascular underpinnings.”
Salk researchers collaborated with scientists at the University of California San Diego on the paper, including co-first author Jiao Zhang and co-senior author John Shyy, among others.
While the findings themselves aren’t entirely a surprise, the paper provides clear confirmation and a detailed explanation of the mechanism through which the protein damages vascular cells for the first time. There’s been a growing consensus that SARS-CoV-2 affects the vascular system, but exactly how it did so was not understood. Similarly, scientists studying other coronaviruses have long suspected that the spike protein contributed to damaging vascular endothelial cells, but this is the first time the process has been documented.
Pseudovirus Models Reveal Independent Toxicity
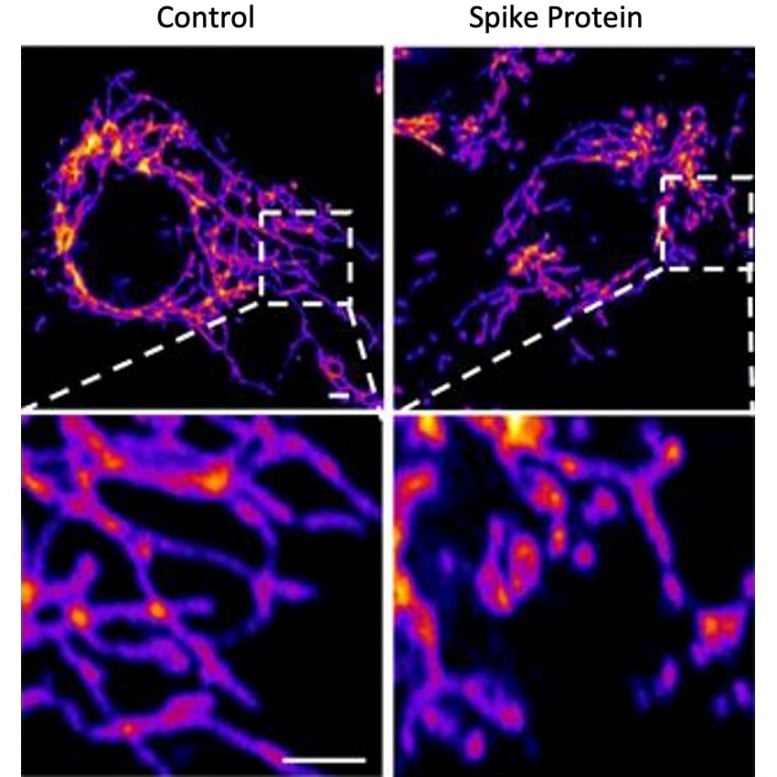
In the new study, the researchers created a “pseudovirus” that was surrounded by SARS-CoV-2 classic crown of spike proteins, but did not contain any actual virus. Exposure to this pseudovirus resulted in damage to the lungs and arteries of an animal model—proving that the spike protein alone was enough to cause disease. Tissue samples showed inflammation in endothelial cells lining the pulmonary artery walls.
The team then replicated this process in the lab, exposing healthy endothelial cells (which line arteries) to the spike protein. They showed that the spike protein damaged the cells by binding ACE2. This binding disrupted ACE2’s molecular signaling to mitochondria (organelles that generate energy for cells), causing the mitochondria to become damaged and fragmented.
Previous studies have shown a similar effect when cells were exposed to the SARS-CoV-2 virus, but this is the first study to show that the damage occurs when cells are exposed to the spike protein on its own.
Mitochondrial Disruption
“If you remove the replicating capabilities of the virus, it still has a major damaging effect on the vascular cells, simply by virtue of its ability to bind to this ACE2 receptor, the S protein receptor, now famous thanks to COVID,” Manor explains. “Further studies with mutant spike proteins will also provide new insight towards the infectivity and severity of mutant SARS CoV-2 viruses.”
The researchers next hope to take a closer look at the mechanism by which the disrupted ACE2 protein damages mitochondria and causes them to change shape.
Reference: “SARS-CoV-2 Spike Protein Impairs Endothelial Function via Downregulation of ACE 2” by Yuyang Lei, Jiao Zhang, Cara R. Schiavon, Ming He, Lili Chen, Hui Shen, Yichi Zhang, Qian Yin, Yoshitake Cho, Leonardo Andrade, Gerald S. Shadel, Mark Hepokoski, Ting Lei, Hongliang Wang, Jin Zhang, Jason X.-J. Yuan, Atul Malhotra, Uri Manor, Shengpeng Wang, Zu-Yi Yuan and John Y-J. Shyy, 31 March 2021, Circulation Research.
DOI: 10.1161/CIRCRESAHA.121.318902
Other authors on the study are Yuyang Lei and Zu-Yi Yuan of Jiaotong University in Xi’an, China; Cara R. Schiavon, Leonardo Andrade, and Gerald S. Shadel of Salk; Ming He, Hui Shen, Yichi Zhang, Yoshitake Cho, Mark Hepokoski, Jason X.-J. Yuan, Atul Malhotra, Jin Zhang of the University of California San Diego; Lili Chen, Qian Yin, Ting Lei, Hongliang Wang and Shengpeng Wang of Xi’an Jiatong University Health Science Center in Xi’an, China.
The research was supported by the National Institutes of Health, the National Natural Science Foundation of China, the Shaanxi Natural Science Fund, the National Key Research and Development Program, the First Affiliated Hospital of Xi’an Jiaotong University; and Xi’an Jiaotong University.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
64 Comments
Some one in the scientific community must comment on the joint sites of cooperation
of the research and the varied nationality of the scientists working on the project!
And what about those many numbers in the total population who contract the virus, recover with seemingly little damage if any to their vascular system? Are their other unknown factors that give them natural immunity?
William, it’s been known since early in the pandemic that maybe 50% of people are not susceptible (that is, who don’t have visible symptoms). However, there seems to be no way to predict who that would be. Nor is it known if one can have no visible symptoms but still have blood related disease that is not visible, or perhaps is not recognized for years.
My assumption is that it depends on the number of virus particles the person receives within a certain period.
If you get in a close contact with someone who has already contracted the virus and spend hours together (like in a hospital room or a small office) there’s a high chance for both to replicate and exchange the virus back and forth more and more exponentially and overloading your bodies before their immune systems respond. An open space or just an open window could reduce the virus transmission. You walk in the park and an infected person coughs near you, so you manage to get few virus particles but keep walking and breathing fresh air. Those particles slowly replicate and start sending signals to your body’s immune system, so it manages to prepare and respond to the infection before it takes you down or even disappear unnoticed.
My other assumption is about the new virus variants: the ones that draw attention do not come from the second most populated country with very high population density – India, as it should be in theory. Most of them have appeared in countries with multiracial populations, though. The miscegenation seems to be the common factor for the number of new variants from South Africa, Brazil, United Kingdom and the United States. The mixed DNA from all races might have given the virus during its replication “a key” for better transmission to any race.
I’m wondering what this research implies about mRNA vaccines that stimulate the production of spike proteins. Would these vaccines not cause similar harm?
It would be nice for them to address it, but given millions vaccinated, apparently not
I feel like there are a lot of propagandists showing up in the comments here. They say things like “vaccines are perfectly safe” which everyone knows is a blatant lie. Vaccination is a calculated risk and with tried and true vaccines the risks are very rare, but no vaccine is “safe” in that deaths and injuries occur due to allergies, negligence and medical incompetence. Anyone who says there is “no risk” with vaccines is a liar, else why would all throughout history there have been huge billion-dollar payouts for vaccine deaths and injuries? That being said, most vaccines are essential for health but with these covid vaccines there has been a suspension of scepticism and government and corporate coercion that is extremely alarming.
Honestly i think you have a bigger problem to worry about then the vaccine Bailey
Your comment is exactly the type of thing that fails the smell test.
To all those wondering if the MRNA vaccine produces the spike.
1. the vaccine has an imprint (kind of like a foot print but not the foot) of the spike and an enzyme which your immune system’s beta cells identify as an antigen it needs to examine.
2. your beta cells then process and read the enzyme with the mrna of the spike to watch out for the foot that will cause all this damage.
Unless the MRNA vaccine gets into one of your cells exactly while its splitting, past the cell wall, past the cell nucleus, guide its self into the exact spot it can attach its self to your cell’s own RNA (when dividing)… its not going to cause any harm to your body. Even if breaks from the enzymes which tell your immune system not to get rid of the useless mrna the body will most likely just get rid of the mrna.
Your beta cells in your immune system know better than to copy random mrna in the body. If the mrna wiggles free from the enzyme that stabilizes the mrna vaccine, the immune system will just eradicate the mrna anyways.
You retards fell asleep in biology 101 of high school and it shows. You dumb asses also watched too much resident evil and think thats actual science and it shows.
I hope in the future humanity will be a little smarter but seeing how the future is based off you, we are F-ed.
“If the mrna wiggles free from the enzyme that stabilizes the mrna vaccine”
What is the enzyme that stabilizes the mRNA vaccines? No such thing is disclosed as being part of the mRNA vaccines, yet this comment of yours is calling others dumb.
Cellular biology 101. Can not happen as this paper suggests. Spikes created inside cells from mRNA are degraded before they exit the cell through the MCH mechanism and the result it the standard immune system hand off is made to fully consume and degrade these protein peptides. Watch this https://youtu.be/9EfToFXwx98?t=479
Wrong. The cells present the spike protein on the surface. When the cell dies, the spike protein is released.
https://www.nytimes.com/interactive/2020/health/moderna-covid-19-vaccine.html
Wrong. He posted a good link. Watch the video. It explains why nothing is “released”
https://www.youtube.com/watch?v=9EfToFXwx98
This preprint suggests that the S1 subunit of the spike protein can remain persistent in non-classical monocytes after vaccination: https://www.researchsquare.com/article/rs-1844677/v1
Circulating spike protein was detected in some cases of post-vaccination myocarditis: https://www.ahajournals.org/doi/10.1161/CIRCULATIONAHA.122.061025
Your statement is inaccurate, because the mRNA vaccines encode for the spike protein to be synthesized and displayed by cells. The spike protein is presented on the surface of the cells, not just individual peptides from a degraded spike protein. Normally, they are attached to cell membranes, but action against the cell by T or NK cells, and/or furin cleavage resulting in different parts of the spike protein separating, can change this.
Is the ‘spike’ protein that allegedly attacks the vascular system the same one that the Pfizer mRNA vaccine stimulates the body to produce? — asking for a concerned friend who took the vaccine.
The vaccine has a limited number of spike proteins, and since it’s not part of a virus it cannot reproduce. The vaccine allows the body to recognize spike proteins so it can learn to fight them, without the risk of a virus that can replicate spike proteins to the point of killing a person.
This explains why the vaccine can make you a little bit ill. If it’s comforting, a little back-of-the-envelope math shows that people are at least 50,000 times more likely to die from covid than to get a serious complication from the vaccine.
How can you give the odds of someone dying of covid vs a vaccine like AstraZenica when you don’t even know their medical history or current medications?
Show the data which proves a healthy 10 year old is 50000 times more likely to die from covid 19 vs a vaccine. The majority of people wouldn’t even know they had covid. Your mis-information is just as bad as some antivaxxers.
Watch this explanation. mRNA created Spikes can not free float around in your body. https://youtu.be/9EfToFXwx98?t=479
Even if it’s turns out to be true that mRna spike proteins can harm your cells. (a pretty tall order) There is not enough of the viral matter in the vaccine to do significant harm without replication, which it cannot do.
These findings should be VERY WIDELY reported to help vaccine skeptics understand the risks they are taking with their own health and that of others.
I would argue the opposite. If spike proteins are dangerous, why do the vaccines produce them?
In reality, Covid-19 is BOTH a vascular and a respiratory disease. For proof, see for instance https://www.aljazeera.com/news/2021/5/3/india-covid-cases-near-20-million-uk-to-send-more-aid-live-news
Don’t the mRNA vaccines produce spike proteins?
Yes, and that’s why it can make you a little ill. But since it’s not part of a virus, it cannot replicate. This gives your body the opportunity to learn how to fight it without the risk of the virus replicating so much that it does serious damage or kills you.
Covid makes most people “not ill at all” or “a little ill” as well. The point of the mRNA vaccines that you don’t *need* the virus itself there to replicate because the mRNA is there to be read and the spike protein is produced. The point of the article is that the spike protein is dangerous, so if the vaccine is producing spike proteins, it’s quite a leap in logic to say that the vaccine is “safe” (whatever definition that is today).
‘Covid makes most people “not ill at all” or “a little ill” as well.’
No one knows that. Most people don’t show symptoms, but no one knows how damaged they body get. Since many cardiovascular diseases are “silent killers”, those people may have their vascular system very damaged and only show any health problem in the future.
Watch this video. mRNA created Spike proteins CAN NOT free float around and infect other AC2 receptors in your body. https://youtu.be/9EfToFXwx98?t=479
Yes, that’s why they induce vascular effects in some cases. Just like the virus.
I agree that if the mRNA vaccines themselves stimulate the human body’s cells to produce spike proteins, shouldn’t there be some concern that by this process the mRNA vaccines could ultimately cause the same vascular damage?
With mRNA vaccine technology the spike protein is made inside the cellular machinery, but it goes on to be destroyed once it is made inside the cells and CAN NOT EXIT the Cell intact.
Confidently incorrect.
You are correct
Vaccines that trigger spike protein production only affect a very limited number of cells, as the genetic material that these vaccines contain does not auto-replicate in the body. In that way, these few cells are able to produce safe levels of spike protein, which are enough to trigger the intended immune response without any of the damaging effects. In contrast, the virus is able to produce many copies of itself in a very short time before the body can make any attempt to control it. This means that very large amounts of circulating spike proteins are produced, generating the adverse effects that the authors identified in their study. If you have access to any vaccine, please take it, they are all good to protect you and your loved ones.
The Pfizer/BioNTech and Moderna vaccines carry instructions to produce a variant of the spike protein that has been locked into what’s called a “prefusion-stabilized conformation”.
Because the vaccine produced spike proteins are locked into this state, they cannot properly bind to the ACE2 receptors in other, healthy cells. This is because the normal spike protein must change its shape in order to achieve “fusion” with the ACE2 receptor.
So the vaccines should be perfectly safe, even in light of this new information.
No vaccine is “perfectly safe.” It is a calculated risk. And the vaccines are in emergency use trials until 2023. Why can’t people sue pharmaceutical companies if they have disabling reactions or their family members die from it? If they are so safe, surely companies should have confidence in their products?
Binding occurs in the pre-fusion state. It is locked in the pre-fusion state to stabilize it not to prevent binding. The spike protein churned out by the body’s cells in response to the mRNA contain a fully-functional s1 binding sub-unit and RBD. The pathogenecity of the S-protein was not yet realized until long after the so-called “vaccine” was created and released.
This research goes against all cellular biology. China researchers trying to scare people from getting mRNA vaccines? Saying you can get just as sick with the Spike protein as the Covd-19 virus itself? Wow. Incredibly ignorant. With nRNA vaccine technology the spike protein is made inside the cellular machinery, but it goes on to be destroyed once it is made inside the cells and CAN NOT EXIT the Cell intact. This is cellular biology 101. Spikes proteins can not be floating around after being made and transit to other AC2 receptors elsewhere in the body. Incredible this document is even published.
If the mRNA vax enters an artery or vein, you’re going to experience a very bad vascular event. Possibly an end-of-life vascular event.
This is not the first paper to show this, Magro (2020) showed this definitively.
A synethetic mRNA gene is a great way to make spike proteins and cause damage because lipid nano-particles can travel into the brain – whereas the virus does not.
Caveat Emptor.
Adam Mercy
Mercy School | Scientific Media
Check out this UK model, which predicts on page 10 #32 that up to 70% of COVID-19 hospitalizations/deaths this fall will be among the twice vaccinated. (An oddly specific characterization given that some COVID-19 vaccinations that do not require a second shot.) See: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/975909/S1182_SPI-M-O_Summary_of_modelling_of_easing_roadmap_step_2_restrictions.pdf
The model anticipates a certain amount of vaccine failure in the 50+ group but then goes on to state on page 18 #56 that the rate of hospitalization/death is NOT evidence that vaccine effectiveness is in doubt but rather a reflection of the fact that a high percentage of people in this age group were fully vaccinated to begin with.
My first instinct would be to blame any Fall/Winter COVID-19 spike on the fact that vaccine-induced immunity is temporary — with some estimates as little as 6-9 months (the apparent hope, however, is that we can go 12 months between boosters). My second instinct would be to blame any Fall/Winter surge in the UK — and similarly here — on variants. However, the model does not account for variants. Given this, what other explanation can there be apart from a lower rate of vaccine effectiveness than we have been led to believe?
Indeed, there are those who have looked at the vaccine clinical trial data and made the observation that the rate of COVID-19 cases among the control group was too small relative to the reduction of cases in the vaccinated group to extrapolate a high rate of effectiveness for vaccines.
What’s more, we have Dr. Fauci and others — President Biden among them too — insisting that the fully vaccinated continue to wear their masks. Dr. Fauci has openly said that it is unknown if vaccines will prevent viral shedding vs. limiting serious illness. (The chief hope, apparently, is to limit hospitalizations/death not necessarily case case counts, many of which may presumably become asymptomatic following vaccination.) Meanwhile, NPR — not exactly a conspiracy website — has also reported that the vaccines may contribute to more variants (hastening so-called immune escape). See: https://www.npr.org/2021/02/09/965703047/vaccines-could-drive-the-evolution-of-more-covid-19-mutants
Taken together, it looks as if Operation Warp Speed delivered a questionable “vaccine” but to recoup their losses Big Pharma still needs to put the jab into people’s arms even though much more is unknown than known!
My replay is to
Pierre Little | May 3, 2021 at 12:13 pm | Reply
With mRNA vaccine technology the spike protein is made inside the cellular machinery, but it goes on to be destroyed once it is made inside the cells and CAN NOT EXIT the Cell intact.
and
pbar | May 3, 2021 at 10:14 am | Reply
The Pfizer/BioNTech and Moderna vaccines carry instructions to produce a variant of the spike protein that has been locked into what’s called a “prefusion-stabilized conformation”.
Because the vaccine produced spike proteins are locked into this state, they cannot properly bind to the ACE2 receptors in other, healthy cells. This is because the normal spike protein must change its shape in order to achieve “fusion” with the ACE2 receptor.
———————
All nice, but the spike proteins are already IN the cells since they are produced there and the study says: […] but this is the first study to show that the damage occurs when CELLS are exposed to the spike protein on its own.
So the spike proteins don’t need to exit the cells and they don’t need to bind to new cells. They are produced INSIDE cells, right where they want to be. I find this very concerning and I already got my first shot.
Okay they are in the cell… so what? There are no AC2 receptors inside the cell. This is the issue, why create spikes inside cellular machinery? Because it is stopped once made, because the cell recognizes is as foreign protein and send it off for destruction, and hands it’s smashed parts from the endoplasmic reticulum (ER) to the MCH mechanism which hands it off to the T-Cell immune system for removal. This is how the immune system becomes ramped up against these degraded pieces of proteins. The premise of this study is the Spike proteins are attaching to the epithelial cells that line our circulatory system. These spikes are wild versions that are already in the circulatory system from an active infection because they are attached to the membrane holding the nucleocapsid. The mRNA vaccines builds the protein within the safe environment of the host cell structure from the inside out…spikes are not created outside this environment. Which is totally different from the wild version. More confirmation watch this: https://youtu.be/9EfToFXwx98?t=479
Thank you Pierre for your explanation! I’m a layman but I think I got it.
The damage happens when a free spike protein binds to a cell. There is no damage when the spike protein is just inside a cell.
So it’s like the spike protein is a burglar. If a burgler, theoretically, is born inside your house and cannot get out alive and is killed by the residents, there is no damage and no damage in the future at other houses. The damage happens when a burgler breaks in with a pry bar (spike protein bind to a cell and wants to get in).
“The mRNA vaccines builds the protein within the safe environment of the host cell structure from the inside out…spikes are not created outside this environment. Which is totally different from the wild version.”
Do you mean like that the virus is synthesized extracellularly? All proteins are synthesized inside cells, not just some of them.
@ Pierre Little
What leads you to believe that the spike protein will remain in the cell vs. expressed into the surrounding tissue by which not only antibody response will be built up (a good thing) but also attack by NK cells (another aspect of the immune system) and thus pro-thrombotic, pro-inflammatory aftereffects?
From what I understand, it’s not entirely clear where vaccine-induced spike proteins will show up because that would require tissue biopsies that were not done in human subjects. But there are clues provided by COVID-19 infection itself — and the implications are disturbing apart from the usual antivaxxer suspects. See: https://www.regulations.gov/document/FDA-2020-N-1898-0246.
The mRNA vaccination process “hijacks” the cell to encode these proteins so we’re not talking a limited number taken up by the vaccine itself after which no more are produced by the body. See: https://www.jpost.com/health-science/could-an-mrna-vaccine-be-dangerous-in-the-long-term-649253
If these spike proteins are expressed in endothelial cells, which are found in the vascular system, brain and other organs, the protein can presumably damage that tissue in a way that may not be readily apparent. Regardless, studies have apparently found that the spike protein can cross the blood-brain barrier — which is a giant red flag for the decision to target the spike protein for vaccine development on the assumption that it was a passive (harmless) target. See: https://pubmed.ncbi.nlm.nih.gov/33053430/ and https://pubmed.ncbi.nlm.nih.gov/33328624/
In Israel, where vaccination rates are much higher, myocarditis cases are under review for possible association with vaccination. Meanwhile, the Department of Justice is investigating myocarditis (inflammation of the heart) following cases in vaccinated service members. Simply put, there are too many VAERS adverse event reports to pass off all serious aftereffects as coincidence or to dismiss them as “background” rates. For example, J&J isn’t the only vaccine where blood coagulation disorders have occurred. We’re on the receiving end of a very incomplete picture from mainstream media because they don’t want to encourage vaccine hesitancy. See: https://www.dailymail.co.uk/news/article-9366963/Coronavirus-Pfizers-Covid-vaccine-linked-blood-clots-AstraZenecas-UK.html
As the old saying goes, where there is smoke there is fire. Does that mean people who are vaccinated face the same health risks as COVID-19 itself? No. Having said that, a more complete answer would depend on the age and health status of the vaccinated individual. In elderly patients who are not already on death’s door, tolerance appears to be quite good (less, even, of flu-like symptoms post vaccination). And since elderly people are at increased risk of COVID-19 complications, it makes more sense for them to be vaccinated than not. But does that hold true for the rest of us?
My educated guess is this: Now that the vaccines are about to be used in kids as young as 12, I expect a more robust immune response and the presence of a better (faster) metabolism will translate into more immediate reports of adverse events among younger vaccine recipients — the same group (with some exceptions!) who would not be expected to be at high risk from COVID-19 itself. In contrast, middle-aged adults with co-morbid conditions who are vaccinated may be more likely to experience delayed adverse events (on the whole).
Of course, none of this even begins to account for how vaccines will alter our immune response, for better or for worse, to COVID-19 variants. A study out of Israel implies that prepping the immune system to expect the original COVID-19 may paradoxically put it at a disadvantage when exposed to the South African variant. Here is a quote “the prevalence of the South African strain among vaccinated individuals who were infected despite their inoculation was eight times higher than its prevalence in the unvaccinated infected population”. See: https://www.timesofisrael.com/real-world-israeli-data-shows-south-african-variant-better-at-bypassing-vaccine/
The COVID-19 vaccine argument thus far has been one-size-fits all: COVID-19 vaccinations are harmful (antivaxxers) vs. helpful (the majority). In reality, the jury is still out — which is why these are approved for emergency use, not “FDA approved” in the usual (year’s long) tradition.
LOL
We are slowly getting to the right conclusion. The solution for the problem with Covid19 is Zyesame/Aviptadil. This is an a synthetic form of Human Vasoactive Intestinal Polypeptide (VIP). This is concentrated in the lungs to help you to breath. This helps to stop the cytokine storm in the beginning and repairs your lungs (if not to damaged). See the next video and it becomes all clear.
https://youtu.be/t9VObXY8szc
https://clinicaltrials.gov/ct2/show/NCT04311697
Aviptadil is now in internatial research project with Remdesivir, which could help in start of the sickness.
https://www.nih.gov/news-events/news-releases/clinical-trial-therapeutics-severely-ill-hospitalized-covid-19-patients-begins
Reading these comments Jonathan is wrong. The new technology treatments give mRNA to teach your own cells to make the spike proteins. The body then recognises these as foreign and starts to make antibodies.
With this in mind, your own body making the spike protein cant be a good thing, however long they last for.
It is not true that spike proteins can’t exit cells, because reactions in your cells do not happen immediately. The mrna enters a cell (also in the brain, thanks to the lipid hull). Much mrna will be destroyed fast by enzymes but some will trigger the cell to produce the spike proteins. Other enzymes destroy the spike proteins and parts of it will be presented to the immune system outside the cell. So far, your assumption is true. BUT: Then the immune system will destroy that cell (also some brain cells which will not regenerate). Now, when the cell gets destroyed, all spike proteins which were produced shortly and did not become destroyed yet will float through your tissue and if you are unlucky, enter a blood vessel. I think, adverse effects of the vaccine happen more often if the needle hits near a bigger blood vessel in the muscle. The vaccines are clearly beneficial to elderly people but I would never let my children be vaxxed without long term studies.
In order to overcome the viruses. It must first be understood by all. Where,when,and how, the virus entered the human biological systems. Who committed such evil ? Why did he or she do it ? Because apparently they don’t believe in GOD AND ANGELS. But Science proves as well as they are proof of INTELLIGENCE EXISTENCE. MAN WAS CREATED BY GREAT INTELLIGENCE. THUS VIRUSES WERE CREATED WITH CERTAIN INTELLIGENCE IN THE PHYSICAL ENVIRONMENT. Somewhere over in China. Or more exactly between Russia,China, and the Eastern Province of Tibet. Lies tall mountains in which hidden within them lies a sea of water. So clean,white,and pure. That if one drink from it ! He becomes clean !!!! And white !!!! Also you become able to look down into the middle of the earth. And look into the heavens and see the sun and all the stars in Heaven. So solving the issues of viruses and other harmful parasites.
Can be overcome by locating this sea.There is one man who saw this sea. And he thought it was glass laying down on the ground. Until he approached it and looking down saw down into the earth and was alarmed. It was as he saw as if someone sliced all the way to the middle. Anyway after drinking this water.He got a different blood type than anyone on earth. And he can transfuse wiping away this virus. Find him.
There are probably millions of viruses, not all of which were deliberately designed by someone. There’s something known as evolution, which applies to viruses in addition to organisms.
The Garden of Eden was found but no one was,or is able to walk into it. It is a beautiful forest with trees so tall and wide. And it is located upon a plateau. Those who are living in the space station over a year. Had to look down and seen it ! There are animals living in it. But the devil and his seed cannot go into it !!!! :
The study was done by Salk. This article is a reprint of what I assume is the original source here: https://www.salk.edu/news-release/the-novel-coronavirus-spike-protein-plays-additional-key-role-in-illness/
So if I took a vaccine, those spike proteins would bypass the first layers of my immune system and will do their damage right in the core of my body. No, thanks.
This study links to an earlier one also sponsored by NIH suggesting that spike protein alone (introduced by vax)can trigger cell signaling and cell host damage e.g. vascular wall thickening the hallmark of Pulmonary artery hypertension. Word of caution re long term safety and giving the vax to young people – The PAH survival is 3 years.
I’m one of those skeptical regarding the vaccines, yet I took the first dose of Pfizer. Do I regret it? Yes. Can I reverse it? No.
I also got a history of rare autoimmune disease involving the blood vessels.
The question is, do these spike proteins stay in the cells long enough to produce any damage ? How long exactly, do Pfizer protocol cover this? what is the ‘half -life’ or the time scale the mRNA and the proteins will be floating around until mRNA is degraded and the spike proteins are met by the immune system? https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7827936/#:~:text=SARS%2DCoV%2D2%20uses%20its%20viral%20membrane%20fusion%20protein%2C,(ARDS)%20%5B5%5D.
I am reading and learning from the comments and knowledge above. I am familiar only in part with some of the details you explain.
I wonder in the optimistic scenario, which I want to believe, then what triggered the – not to many but enough to ask – cases of thrombocytopenia? that happened with all Covid (and other) vaccines in use today? And I understand some clots as well, including with the Pfizer vaccine.
I am asking this also because I want to get vaccinated but just like the previous comment with the autoimmune condition, I have had an autoimmune condition, the hives on and off for the past 15years (I am not that young anymore though), cause unknown although stress and some chemicals make them worse. I am not as much worried about anaphylaxis but I read that the latest discoveries and treatments for hives involve anti-coagulants regarding a ‘thrombin factor’ level. I don’t know if this should worry me more or the same (?).
https://www.euronews.com/2021/06/02/pfizer-covid-19-vaccine-linked-to-myocarditis-in-young-men-says-israel-s-health-ministry
Heart inflammation from Vaccine discovered …
Most, if not all of you people are idiots – you just want to be heard and want to rant pseudoscience, and no where remotely close. Did anyone of you develop those vaccines? No, so learn a lesson from a dummy, “Don’t trust pharmaceuticals’ and governmental joint campaigns!” They’ve already poisoned the water, the food and the air, not to mention taxing you for everything – so, yeah, another vaccine campaign because they just care so much about the cows…wake up you fools!
What a bunch of unintelligible nonsense. I’m not even sure what your point is.
Those who point out that the vaccine spike proteins can enter the bloodstream are correct. This is because the spike parts are displayed on the cell. (This also results in antibody production as others are correctly pointing out.). However, the displayed parts also cause the innate immune system to attack and destroy the cell wall. This frees the (likely many) whole and partial spike proteins inside the cell into the blood. This is how they escape without being torn to pieces. Which process(es) dominate will depend on each individuals’ particular immune system.
the spike protein induced by the vaccine are made in the cell – if they stayed in cell or were destroyed there = no antibodies would be made. So they are designed to migrate and attach on the outside of that cell so the cells in the immune system can notice them and create antibodies. Other parts of the immune system such as killer Tcells will destroy the spike protein “infected” cell with its exterior attached spike protein. Before it is destroyed it causes micro clots if the spike producing cell is part of the endothelial cell wall. This has been shown to happen by researchers or doctors perfforming a D Dimr test on recently vaccinated people. The vaccines were supposed to be staying in the arm where the recipient was jabbed but this is now known to be false. They travel extensively through the lymph system and a large number end up in the spleen. The mRNA lipid coated vesicles (numbering in the trillions from the jab also enter the blood stream where they travel the whole body even past the blood brain barrier. Any cell they successfully enter will then make trillions of the vaccine induced synthetic (made on an altered computer model of the protein) spike protein. I imagine the commenter who thought that spike making cells destroyed would release unnattached spike protein into the body was correct. It is now known that many of the toxid effects of being infected by SARS COV 2 stem directly from the spike protein – ie the spike protein itself is a pathogen. To use a vaccine that induces a person’s cells to manufacture a pathogen in the trillions seems foolish on the face of it.
I just listened to a simplified lecture on why the spike proteins are mostly destroyed inside the cell as some of the pro-vacciners stated. Even that professor agreed that the cell will usually be destroyed by the immune system releasing intact spike proteins. It also means that while naturally acquired immunity from the infection of SARS COv 2 will have targets from all different parts of the virus, the antibodies produced by the vaccine will only be active for small fragments of the transfigured spike protein induced by the vaccine mRNA. My statement that the spike protein migrates to the surface of the cell so as to be recognized by the immune system is not a very accurate simplification. Only small fragments of the ubiquoitim – proteosome protein degradation process of the induced spike protein are presented on the surface of the cell by the MHC 1 ANTIGEN presentation system. It has been stated by Dr Hoffee and Dr Mercola that something like 40 trillion lipid coated MRNA are injected by each jab. Each of those are thought to induce more than 2000 spike proteins in the cells they enter. Every cell invaded by the vaccine mRNA will eventually be destroyed by the immune system releasing spike protein. As mentioned, the spike protein is itself a pathogen. What could possibly go wrong?