Research shows TTK21 promotes neuron regrowth in mice with spinal injuries, with increased axon sprouting suggesting future therapeutic potential for spinal cord injury patients.
Currently, there are no effective therapies for spinal cord damage; physical therapy may help patients recover some movement, but the results are severely limited in severe cases due to the inability of spinal neurons to regenerate naturally after injury.
However, in a study that was recently published in PLOS Biology, scientists led by Simone Di Giovanni at Imperial College London in the UK demonstrate that when given to mice 12 weeks after a serious injury, weekly treatments with an epigenetic activator can encourage the regrowth of sensory and motor neurons in the spinal cord.
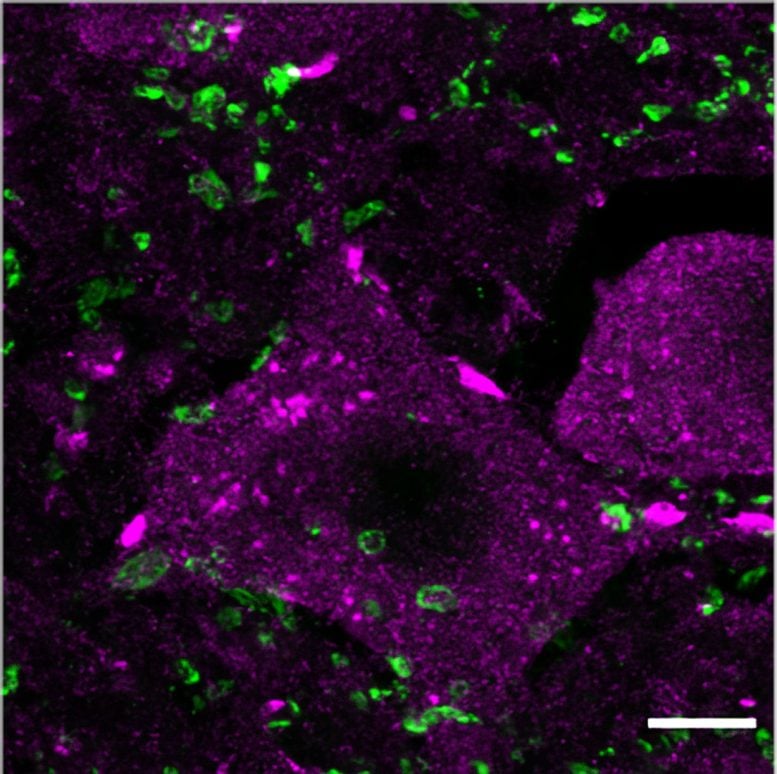
Activating Regrowth with Epigenetic Therapy
Continuing their past success, the researchers utilized a tiny molecule called TTK21 to activate genetic programming that induces axon regeneration in neurons. TTK21 affects the epigenetic state of genes by activating the CBP/p300 family of coactivator proteins. They tested TTK21 treatment in a mouse model of severe spinal cord injury. The mice were raised in an enriching environment that encouraged them to be physically active, as is recommended for human patients.
The treatment started 12 weeks after the severe spinal cord injury and lasted 10 weeks. Researchers found several improvements after TTK21 treatment compared with the control treatment. The most apparent effect was increased axon sprouting in the spinal cord. They also discovered that motor axon retraction above the point of injury halted and sensory axon growth increased.
These changes were likely due to the observed increase in gene expression related to regeneration. The next step will be to enhance these effects even more and to trigger the regenerating axons to reconnect to the rest of the nervous system so that animals can regain their ability to move with ease.
Di Giovanni adds, “This work shows that a drug called TTK21 that is administered systemically once/week after a chronic spinal cord injury (SCI) in animals can promote neuronal regrowth and an increase in synapses that are needed for neuronal transmission. This is important because chronic spinal cord injury is a condition without a cure where neuronal regrowth and repair fail. We are now exploring the combination of this drug with strategies that bridge the spinal cord gap such as biomaterials as possible avenues to improve disability in SCI patients.”
Reference: “CBP/p300 activation promotes axon growth, sprouting, and synaptic plasticity in chronic experimental spinal cord injury with severe disability” by Franziska Müller, Francesco De Virgiliis, Guiping Kong, Luming Zhou, Elisabeth Serger, Jessica Chadwick, Alexandros Sanchez-Vassopoulos, Akash Kumar Singh, Muthusamy Eswaramoorthy, Tapas K. Kundu and Simone Di Giovanni, 20 September 2022, PLOS Biology.
DOI: 10.1371/journal.pbio.3001310
The study was funded by the International Spinal Research Trust, Marina Romoli Onlus, the Rosetrees Trust, and the Brain Research Trust.
The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.