A Caltech study identified trunk neural crest cells as the origin of protective bony scales in fish, shedding light on the evolution of vertebrate armor.
About 350 million years ago, your evolutionary ancestors—and the ancestors of all modern vertebrates—were merely soft-bodied animals living in the oceans. In order to survive and evolve to become what we are today, these animals needed to gain some protection and advantage over the ocean’s predators, which were then dominated by crustaceans.
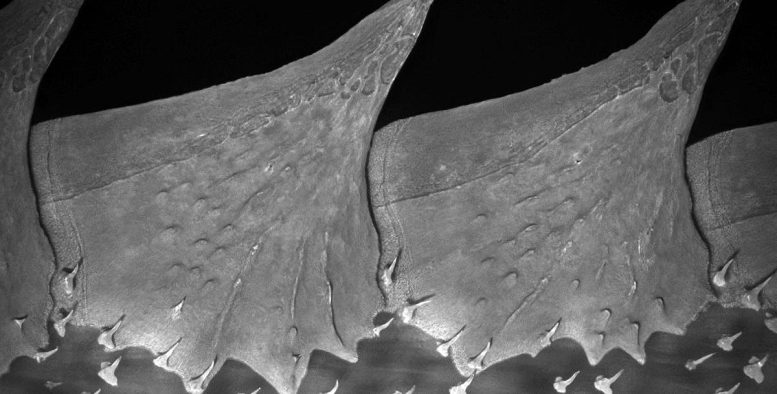
The evolution of dermal armor, like the sharp spines found on an armored catfish or the bony diamond-shaped scales, called scutes, covering a sturgeon, was a successful strategy. Thousands of species of fish utilized varying patterns of dermal armor, composed of bone and/or a substance called dentine, an important component of modern human teeth. Protective coatings like these helped vertebrates survive and evolve further into new animals and ultimately humans.
But where did this armor come from? How did our ancient underwater ancestors evolve to grow this protective coat?
New Research Identifies the Source of Bony Armor
Now, using sturgeon fish, a new study finds that a specific population of stem cells, called trunk neural crest cells, are responsible for the development of bony scutes in fish. The work was conducted by Jan Stundl, now a Marie Sklodowska-Curie postdoctoral scholar in the laboratory of Marianne Bronner, the Edward B. Lewis Professor of Biology and director of the Beckman Institute at Caltech. A paper describing the research was published recently in the journal Proceedings of the National Academy of Sciences (PNAS).

The Bronner laboratory has long been interested in studying neural crest cells. Found in all vertebrates including fish, chickens, and ourselves, these cells become specialized based on whether they arise from the head (cranial) or spinal cord (trunk) regions. Both cranial and trunk neural crest cells migrate from their starting points throughout the animal’s developing body, giving rise to the cells that make up the jaws, heart, and other important structures. After a 2017 study from the University of Cambridge showed that trunk neural crest cells give rise to dentine-based dermal armor in a type of fish called the little skate, Stundl and his colleagues hypothesized that the same population of cells might also give rise to bone-based armor in vertebrates broadly.
To study this, Stundl and the team turned to the sturgeon fish, specifically the sterlet sturgeon (Acipenser ruthenus). Modern sturgeons, best known for their production of the world’s most expensive caviar, still have many of the same characteristics as their ancestors from millions of years ago. This makes them prime candidates for evolutionary studies.
Tracking the Development of Bony Scutes
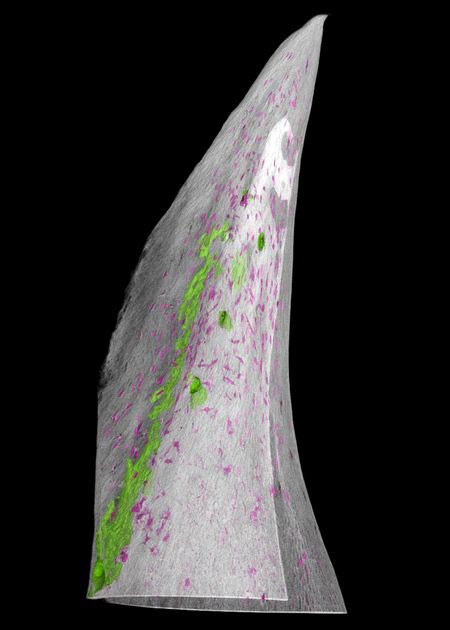
Using sturgeon embryos grown at the Research Institute of Fish Culture and Hydrobiology in the Czech Republic, Stundl and his team used fluorescent dye to track how the fish’s trunk neural crest cells migrated throughout its developing body. Sturgeons begin to develop their bony scutes after a couple of weeks, so the researchers kept the growing fish in a darkened lab in order to not disturb the fluorescent dye with light.
The team found fluorescently labeled trunk neural crest cells in the exact locations where the sturgeon’s bony scutes were forming. They then used a different technique to highlight the fish’s osteoblasts, a type of cell that forms bone. Genetic signatures associated with osteoblast differentiation were found in the fluorescent cells in the fish’s developing scutes, providing strong evidence that the trunk neural crest cells do in fact give rise to bone-forming cells. Combined with the 2017 findings about neural crest cells’ role in forming dentine-based armor, the work shows that trunk neural crest cells are indeed responsible for giving rise to the bony dermal armor that enabled the evolutionary success of vertebrate fish.
“Working with non-model organisms is tricky; the tools that exist in standard lab organisms like mouse or zebrafish either do not work or need to be significantly adapted,” says Stundl. “Despite these challenges, information from non-model organisms like sturgeon allows us to answer fundamental evolutionary developmental biology questions in a rigorous manner.”
“By studying many animals on the Tree of Life, we can infer what evolutionary events have taken place,” says Bronner. “This is particularly powerful if we can approach evolutionary questions from a developmental biology perspective, since many changes that led to diverse cell types occurred via small alterations in embryonic development. We were very fortunate to receive funding from Caltech’s Center for Evolutionary Sciences, which helped us make studies of this sort possible.”
Caltech’s Center for Evolutionary Science (CES) is an Institute-wide, multi-division organization that recognizes and supports the investigation of evolutionary change in the natural world via both biotic and anthropogenic forces.
“Evolution is a central theme that runs through all of biology; it unifies our discipline,” says Joe Parker, Assistant Professor of Biology and Biological Engineering, Chen Scholar, and co-director of the CES. “Caltech is an incredible place with so many groups pursuing evolutionary problems in different contexts, including at the interface of evolution and development biology—as this study so beautifully shows.”
The paper is titled “Ancient vertebrate dermal armor evolved from trunk neural crest” by Jan Stundl, Megan L. Martik, Donglei Chen, Desingu Ayyappa Raja, Roman Franěk, Anna Pospisilova, Martin Pšenička, Brian D. Metscher, Ingo Braasch, Tatjana Haitina, Robert Cerny, Per E. Ahlberg and Marianne E. Bronner, 17 July 2023, Proceedings of the National Academy of Sciences.
DOI: 10.1073/pnas.2221120120
In addition to Bronner and Stundl, Caltech co-authors are former postdoctoral scholar Megan Martik, now at the University of California Berkeley, and postdoctoral scholar Desingu Ayyappa Raja. Additional co-authors are Donglei Chen, Tatjana Haitina, and Per Ahlberg of Uppsala University in Uppsala, Sweden; Roman Franěk and Martin Pšenička of the University of South Bohemia in the Czech Republic; Anna Pospisilova and Robert Cerny of Charles University in Prague, Czech Republic; Brian Metscher of the University of Vienna in Austria; and Ingo Braasch of Michigan State University. Funding was provided by the European Union’s Horizon 2020 research and innovation program; the National Institutes of Health; a Wallenberg Scholarship from the Knut & Alice Wallenberg Foundation; the Helen Hay Whitney Foundation; the Ministry of Education, Youth and Sports of the Czech Republic; the Czech Science Foundation; and the National Science Foundation.
Bronner and Parker are affiliated faculty members with the Tianqiao and Chrissy Chen Institute for Neuroscience at Caltech.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.