Scientists from Yale University have figured out a way to refine bulk metallic glasses to improve their electrochemical performance.
Results of the research, based in the lab of chemical and environmental engineering professor André D. Taylor, are published in the December 22 issue of Advanced Materials.
Bulk metallic glass (BMG), also known as amorphous metal alloy, exhibits superior mechanical properties and great formability under low temperature compared with general alloys. Numerous efforts have been applied to develop these materials for biomedical devices, high-efficiency transformers, and for products that require high-strength materials, such as golf clubs.
Working with the lab of Yale professor Jan Schroers, Taylor pioneered the use of BMGs as catalysts in fuel cells. BMGs have the strength of metal, but can be shaped like a plastic, so they can be used to form nanowires and achieve high-surface areas. Increasing surface area leads to an increased performance in current density because all of the electrochemical reactions are on the surface.
To build on this research, Taylor’s lab set out to develop a way to refine the materials, using subtractive and additive processes. Unusual in its scope, the 17-author paper is the result of a few years of research and work within the Yale NSF MRSEC Center for Research on Interface Structures and Phenomena (CRISP). Yale Ph.D. student Jinyang Li, General Motors researcher Ryan C. Sekol, and University of Campinas professor Gustavo Doubek are the co-lead authors of the paper. Sekol is a former member of Taylor’s lab and Doubek is a visiting fellow at the lab.
“In our first generation, we were able to demonstrate high surface areas using the high formability of metallic glass,” Li said. “For this second generation, we wanted to push this even further using subtractive and additive processes.”
The additive process uses either what’s known as under-potential deposition or galvanic displacement to add materials to a BMG. For example, a combination of platinum and ruthenium is an ideal catalyst for certain applications, but you can’t make a metallic glass with it.
“We’re showing that you can take a very good, high-formability metallic glass, create nanowires and then add ruthenium to it later,” said Taylor, who likened the work to that of the Catalan artist Joan Míro, who challenged conventional uses of materials and processes. “Now you’re not limited to the glass-forming ability of your metals.”
Taylor said that other materials can be added using the same process. Platinum has no pseudocapacitive properties, which allow a material to store electrical charge, so they added manganese dioxide.
“It’s a cheap material, but has a high performance for pseudocapacitors,” Taylor said.
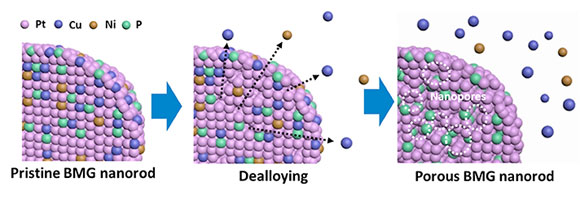
With the subtractive process, Taylor said, researchers can eliminate certain materials from a multi-component BMG, a process known as dealloying. Less noble metals, which don’t act as catalysts, are leached from the surface. Using this technique, high surface areas can be formed from solid structures or nanowires can be converted into branched dendritic structures through electrochemical cycling.
Reference: “Guided Evolution of Bulk Metallic Glass Nanostructures: A Platform for Designing 3D Electrocatalytic Surfaces” by Gustavo Doubek, Ryan C. Sekol, Jinyang Li, Won-Hee Ryu, Forrest S. Gittleson, Siamak Nejati, Eric Moy, Candy Reid, Marcelo Carmo, Marcelo Linardi, Punnathat Bordeenithikasem, Emily Kinser, Yanhui Liu, Xiao Tong, Chinedum O. Osuji, Jan Schroers, Sundeep Mukherjee and André D. Taylor, 22 December 2015, Advanced Materials.
DOI: 10.1002/adma.201504504
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Increasing surface area leads to an increased performance in current density, because all of the electrochemical reactions are on the surface.