How cancer cells tug against each other determines whether they can migrate elsewhere in the body.
Understanding how cancerous cells spread from a primary tumor is important for any number of reasons, including determining the aggressiveness of the disease itself. The movement of cells into the extracellular matrix (ECM) of neighboring tissue is an essential step in cancer progression that directly correlates to the onset of metastasis.
In a paper published today in APL Bioengineering, by AIP Publishing, a team of researchers from Germany and Spain used a breast cancer cell line panel and primary tumor explants from breast and cervical cancer patients to examine two different cellular contractility modes: one that generates collective tissue surface tension that keeps cell clusters compact and another, more directional, contractility that enables cells to pull themselves into the ECM.
“We focused on two parameters, namely the ability of the cells to pull on the ECM fibers and generate traction forces and on their ability to pull on each other, thereby generating a high tissue surface tension,” said author Eliane Blauth. “We linked each property to different contractile mechanisms and asked how they are connected to cancer cell escape and tumor aggressiveness.”
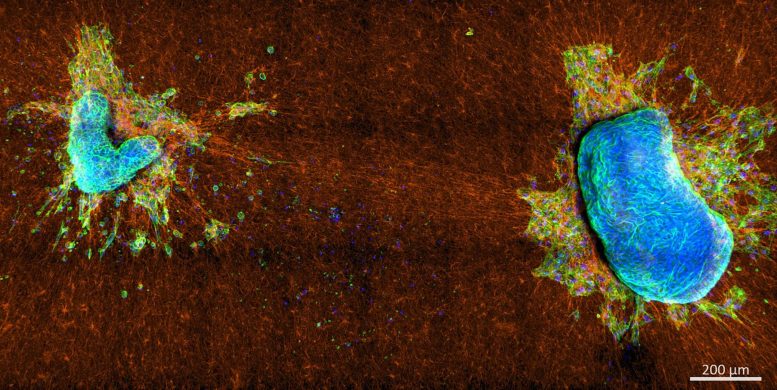
The team found that more aggressive cells pull more strongly on the ECM than on themselves while noninvasive cells pull more strongly on themselves than on the ECM – and that the different pulling behaviors are attributed to different structures of actin cytoskeleton inside the cells. Invasive cells use predominantly actin stress fibers — thick actin bundles that span the cell — to generate forces on their surroundings, while noninvasive cells generate forces through their actin cortex, a thin network directly under the cell membrane.
The study showed it is not the overall magnitude of these contractility modes but the interplay between them that determines a cell’s potential for escape. Experiments with only moderately invasive cells demonstrated the total force these cells generate on the ECM fibers is comparable to that of noninvasive cells, yet they can still detach and invade the ECM, which is not possible for noninvasive cells.
“The noninvasive cells still have a high cortical contractility, keeping them together, while the moderately invasive cells have a nearly disappearing cortical contractility,” said Blauth. “So not much is holding them back even though they pull much weaker on the ECM fibers.”
The team’s measurements with patient-derived vital tumor explants confirmed their findings from the cell line experiments. Here, the number of cells with a high cortical contractility decreased during tumor progression.
“This further indicates that the ability of the cells to pull on each other and hold themselves clustered together becomes weaker as the tumor grows, potentially increasing metastasis risk.”
Reference: “Different contractility modes control cell escape from multicellular spheroids and tumor explants” by Eliane Blauth, Steffen Grosser, Frank Sauer, Mario Merkel, Hans Kubitschke, Enrico Warmt, Erik W. Morawetz, Philip Friedrich, Benjamin Wolf, Susanne Briest, Grit Gesine Ruth Hiller, Lars-Christian Horn, Bahriye Aktas and Josef A. Käs, 7 May 2024, APL Bioengineering.
DOI: 10.1063/5.0188186
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.