These insights could drive the development of new drugs to treat transthyretin amyloidosis, a progressive and fatal disease.
Transthyretin, a small but crucial protein, plays a vital role in transporting hormones through the blood and spinal fluid. However, when it misfolds after secretion, it can lead to serious health problems. Misfolded transthyretin forms toxic clumps in the heart and along nerves, causing transthyretin amyloidosis (ATTR)—a progressive and often fatal disease. ATTR affects up to 25% of men over 80, leading to symptoms such as shortness of breath, dizziness, and numbness or tingling in the extremities.
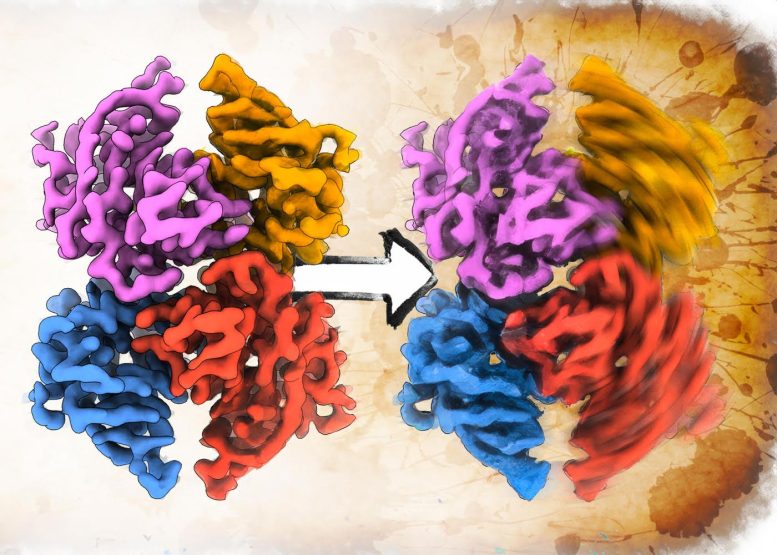
In a breakthrough study, researchers at Scripps Research have revealed new structural insights into transthyretin. Their findings, published in Nature Structural & Molecular Biology, show how the protein’s three-dimensional asymmetry may contribute to its instability. This discovery could pave the way for new drug treatments to combat ATTR.
“We’ve unveiled a molecular complexity that has been hidden from researchers for decades, which enables us to design better medicines to stabilize transthyretin,” says co-senior author Gabriel Lander, PhD, professor at Scripps Research.
“The new structures reveal differences in two thyroid hormone binding sites previously thought to be identical, and help explain why a drug binding to one site changes the ability of drugs to bind the opposing site,” adds Jeffery Kelly, PhD, the Lita Annenberg Hazen Professor of Chemistry at Scripps Research and a co-senior author of the study.
Challenges in Studying Transthyretin’s Structure
To determine the three-dimensional structure of small proteins such as transthyretin, researchers often turn to crystallography, in which proteins are forced into a large, repetitive crystal structure before being imaged. However, the structure of a crystallized protein doesn’t always reflect the conformation of individual free-floating proteins in the body.
Another method, cryo-electron microscopy (cryo-EM), flash-freezes proteins to catch them in their more natural structures. However, these frozen proteins are then suspended in a liquid, and small proteins like transthyretin tend to get stuck at the air-liquid boundary rather than remaining fully submerged. This affects both the proteins’ structural stability and the ability to illuminate their detailed structure.
To overcome this challenge, Lander’s group developed a thin graphene-coated grid to which transthyretin molecules could naturally adhere. Then, they rapidly plunged this grid into liquid ethane to freeze the sample. This process trapped the transthyretin molecules in place on the graphene surface and preserved their natural conformations to mimic how they would appear when moving through the body’s blood or fluids.
“We built on the 2019 work from the Yan lab at Princeton when making our grids. Getting the surface chemistry right is crucial for this type of study. With small proteins like transthyretin, creating a high-quality sample is just the beginning; analyzing the data is also part of the challenge,” says Benjamin Basanta, PhD, a former research associate in the Lander Lab and first author of the new paper.
A Molecular “Breathing” Mechanism and Its Link to Disease
When the team tested the approach with transthyretin, they discovered that transthyretin forms asymmetric structures with two differently shaped binding pockets. Based on the more than 200 crystal structures that had been solved in the past, these binding sites had been assumed to be identical. The researchers showed that this variation was because the transthyretin complex was constantly wiggling between two different states—like a molecular version of “breathing,” according to Lander. This asymmetry in the native structure of transthyretin also presents a hypothesis for how the process of dissociation and misfolding could occur then lead to the clumping of the protein and subsequent disease.
Attaching the ATTR drug tafamidis—developed by the Kelly lab—to one or both of the transthyretin binding sites, they then found, stabilized the molecule, and minimized this movement.
Now, Lander and his colleagues aim to study how this structure and its stabilization relate to ATTR, and how drugs targeting transthyretin could treat the disease. They also say that their graphene grid method could be used to determine the structures of other small and unstable proteins—including the amyloid-beta peptide that builds up in the brain in Alzheimer’s disease.
“The methodologies we’ve developed have opened new doors to avenues of treatment that could one day protect patients from not just TTR amyloidosis, but other amyloid diseases as well,” says Lander.
Reference: “The conformational landscape of human transthyretin revealed by cryo-EM” by Benjamin Basanta, Karina Nugroho, Nicholas L. Yan, Gabriel M. Kline, Evan T. Powers, Felix J. Tsai, Mengyu Wu, Althea Hansel-Harris, Jason S. Chen, Stefano Forli, Jeffrey W. Kelly and Gabriel C. Lander, 22 January 2025, Nature Structural & Molecular Biology.
DOI: 10.1038/s41594-024-01472-7
This work was supported by the National Institutes of Health (GM142196, AG067594, DK046335) and a Postdoctoral Fellowship from the George E. Hewitt Foundation for Medical Research.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.