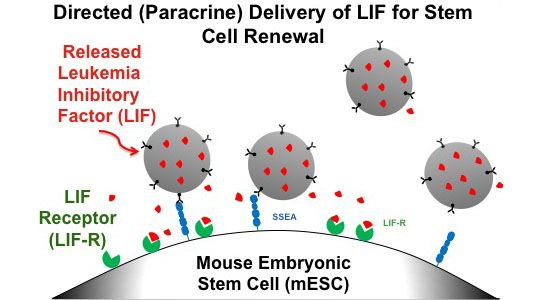
By packaging Leukemia Inhibitory Factor (LIF) inside biodegradable nanoparticles, scientists developed a nanoparticle-based system to deliver growth factors to stem cells in culture, resulting in cell colony growth with a 10,000 fold lower dose of LIF when using the nanoparticle-based delivery system compared to traditional methods using soluble LIF in a growth medium.
Stem cells – unspecialized cells that have the potential to develop into different types of cells – play an important role in medical research. In the embryotic stage of an organism’s growth, stem cells develop into specialized heart, lung, and skin cells, among others; in adults, they can act as repairmen, replacing cells that have been damaged by injury, disease, or simply by age. Given their enormous potential in future treatments against disease, the study and growth of stem cells in the lab is widespread and critical. But growing the cells in culture offers numerous challenges, including the constant need to replenish a culture medium to support the desired cell growth.
Tarek Fahmy, Associate Professor of Biomedical Engineering & Chemical & Environmental Engineering, and colleagues have developed a nanoparticle-based system to deliver growth factors to stem cells in culture. These growth factors, which directly affect the growth of stem cells and their differentiation into specific cell types, are ordinarily supplied in a medium that is exchanged every day. Using the researchers’ new approach, this would no longer be necessary.
“Irrespective of their scale or nature, all cell culture systems currently in practice conventionally supply exogenous bioactive factors by direct addition to the culture medium,” says Paul de Sousa, a University of Edinburgh researcher and co-principal investigator on the paper. With that approach, he explains, “Cost is one issue, especially during prolonged culture and when there is a requirement for complex cocktails of factors to expand or direct differentiation of cells to a specific endpoint.”
A second issue, says de Sousa, is specificity: growth factors supplied by direct addition to the culture medium can lead to the growth of undesired cell populations, which can end up competing with the growth of the desired cell types.
“A relatively unexplored strategy to improve the efficiency of stem cell culture is to affinity-target critical bioactive factors sequestered in biodegradable micro or nanoparticles to cell types of interest,” explains de Sousa, “thereby achieving a spatially and temporally controlled local ‘paracrine’ stimulation of cells.”
Fahmy and his colleagues packaged leukemia inhibitory factor, which supports stem cell growth and viability, inside biodegradable nanoparticles. The nanoparticles were “targeted” by attaching an antibody – one specific to an antigen on the surface of mouse embryonic stem cells being grown in culture. As a result, the nanoparticles target and attach themselves to the stem cells, ensuring direct delivery of the bioactive factors packaged inside.
The researchers have previously demonstrated the potential uses of this approach in drug delivery and vaccination, including targeted delivery of Leukocyte Inhibitory Factor (LIF), which prevents certain types of white blood cells from migrating, in order to regular immune responses. In stem cell cultures, LIF is also the key factor required to keep the cells alive and let them retain their ability to develop into specialized types of cells.
In this research, Fahmy and his colleagues packed LIF into the biodegradable nanoparticles for slow-release delivery to the stem cells in culture. Their results showed cell colony growth with a 10,000 fold lower dose of LIF when using the nanoparticle-based delivery system compared to traditional methods using soluble LIF in a growth medium. While a stem cell culture sustained using a traditional method of exchanging growth medium consumes as much as 25 nanograms of LIF in a day – about 875 nanograms after five weeks of culture – only 0.05 total nanograms of LIF would be required to achieve the same level of growth using the nanoparticle delivery system, a remarkable reduction in the required materials.
The next step is to use these systems with human cells to direct their differentiation into hematopoietic cells—blood products. Clinical and industrial translation of this ability requires efficient and cost effective strategies for cell manufacturing. In principle, this method offers a means to produce standardized or individually tailored cells to overcome challenges associated with donated blood products.
Reference: “Paracrine signalling events in embryonic stem cell renewal mediated by affinity targeted nanoparticles” by Bruna Corradettia, Paz Freilea, Steve Pellsa, Pierre Bagnaninchia, Jason Park, Tarek M. Fahmy and Paul A. de Sousaa, 30 June 2012, Biomaterials.
DOI: 10.1016/j.biomaterials.2012.06.011
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.