The axon initial segment (AIS) is where nerve signals begin, and its adaptability is key to brain function. Scientists have found that the AIS changes in response to activity levels, helping neurons maintain balance. By using advanced imaging techniques, they have observed these changes live for the first time, deepening our understanding of brain plasticity.
The Axon Initial Segment – A Critical Starting Point
Our nerve cells, or neurons, communicate by sending fast electrical signals called action potentials. These signals always begin in a specific part of the neuron known as the axon initial segment (AIS). The AIS is the first section of the axon — the long, thin extension of the neuron that transmits signals to other nerve cells. It acts like a control center, deciding when to start an action potential before it travels down the axon to relay information.
Scientists previously discovered that the AIS is not a fixed structure but can change in response to activity levels in the brain. This adaptability, known as plasticity, allows the brain to adjust its connections and structures to regulate electrical activity — an essential process for learning and memory. When brain activity is high, the AIS can shorten; when activity is low, it can lengthen. However, the exact mechanisms behind these changes and the speed at which they occur were unclear.
Researchers Amélie Fréal and Nora Jamann, working in Maarten Kole’s lab, have now observed this adaptability in real time for the first time. Their groundbreaking work has also identified the molecular processes that drive these changes within the axon.
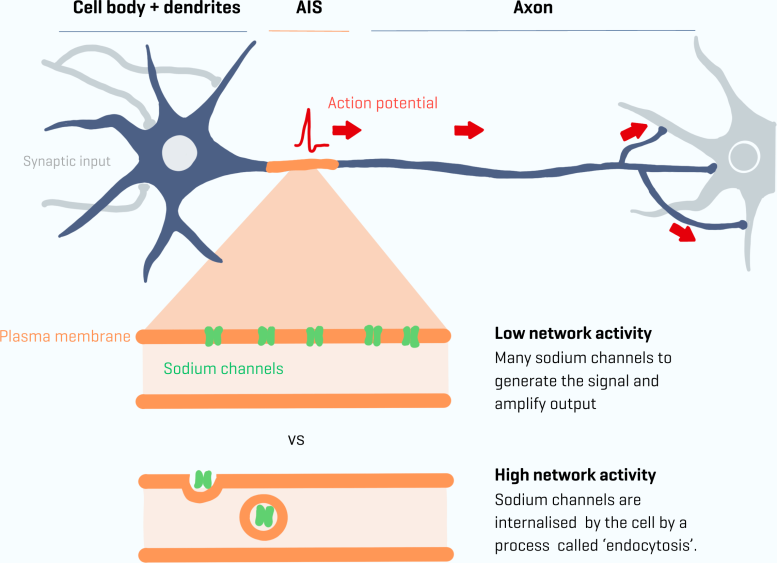
Essential Gates of the Axon Initial Segment
Key players in this process are ion gates located at the segment, also known as sodium channels. The team developed new tools to study these sodium channels and their supporting proteins. They discovered that the number of sodium channels in the cell’s segment can change rapidly, within an hour. This rapid change is mediated by a process called endocytosis, in which the sodium channels are taken up into vesicles within the cell.
Nora Jamann explains, “You can think of this adaptability as a kind of amplifier that allows you to fine-tune the input. The longer the AIS, the less current you need. You can enhance the cell’s output. If this isn’t properly adjusted, learning can be compromised.”
“When you learn, the activity in the neural network constantly fluctuates. Neurons need to operate with extreme variations in levels of activity—both too low and too high activity can be harmful for memory formation.”
Her previous experiments in mice illustrate this adaptability: a mouse with clipped whiskers receives reduced sensory input. As a result, the number of sodium channels in the AIS increases to maintain balance. The opposite also occurs: if there is too much input, such as when the mouse is placed in a new environment with high activity, the AIS becomes slightly shorter and has fewer sodium channels. But how this works and how fast this plasticity occurs have always been fundamental questions for neuroscientists.
Live Imaging Plasticity
Amélie Fréal says, “To address this question, we faced a significant challenge: how can we capture plasticity live? If you wonder how the AIS adapts, you want to actually see it moving. This was not previously possible in the field. In this research, we used two new tools: first, a special mouse model with the AIS labeled with a fluorescent protein, allowing us to observe the mechanism and record the temporal changes in brain slices. Second, we used molecular tools that made sodium channels visible in cell cultures. This made it possible to track the sodium channels live for the first time.”
Jamann continues, “The plasticity we observe in the AIS closely resembles what we know about synaptic plasticity. This is the most well-known form of plasticity and relates to the ability of the connection (the synapse) between two nerve cells to change in strength. Synaptic plasticity is directly linked to learning and memory. But the same mechanism also occurs in the AIS.”
Amélie Fréal adds, “If you want to be a bit provocative, you can even say that a lot of change occurs in the synapse, but only the relevant information is forwarded to the next nerve cell. This decision is made in the AIS, so changes in this region are genuinely important for the cell’s function.”
Collaboration and Future Implications
Fréal concludes, “This study brings together different areas of expertise, which I strongly support in research. Maarten Kole’s lab has all the equipment to record and see the activity, and I came with novel tools to image the nanoscale structures. Together, we have been able to establish that when activity changes, structure changes as well. This observation will help us see plasticity in a broader perspective. When looking at plasticity in general, we now need to consider these changes in the AIS. Through this collaboration, we have been able to combine our strengths to advance knowledge. That’s what I’m most proud of.”
Reference: “Sodium channel endocytosis drives axon initial segment plasticity” by Amélie Fréal, Nora Jamann, Jolijn Ten Bos, Jacqueline Jansen, Naomi Petersen, Thijmen Ligthart, Casper C. Hoogenraad and Maarten H. P. Kole, 15 September 2023, Science Advances.
DOI: 10.1126/sciadv.adf3885
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.