Researchers at the University of Helsinki in collaboration with researchers from Åbo Akademi University (Finland) and Huazhong University of Science and Technology (China) have developed a new anti-cancer nanomedicine for targeted cancer chemotherapy. This new nano-tool provides a new approach to use cell-based nanomedicines for efficient cancer chemotherapy.
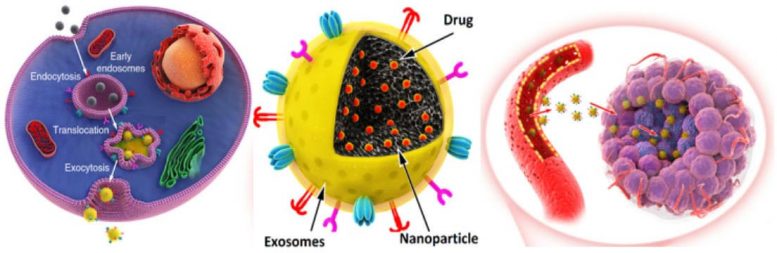
Exosomes contain various molecular constituents of their cell of origin, including proteins and RNA. Now the researchers have harnessed them together with synthetic nanomaterial as carriers of anticancer drugs. The new exosome-based nanomedicines enhanced tumor accumulation, extravasation from blood vessels and penetration into deep tumor parenchyma after intravenous administration.
“This study highlights the importance of cell-based nanomedicines”, says the principal investigator and one of the corresponding authors of this study, Hélder A. Santos, Associate Professor at the Faculty of Pharmacy, University of Helsinki.
Nanoparticles-based drug delivery systems have shown promising therapeutic efficacy in cancer. To increase their targetability to tumors, nanoparticles are usually functionalized with targeted antibodies, peptides or other biomolecules. However, such targeting ligands may sometimes have a negative influence on the nanoparticle delivery owing to the enhanced immune-responses.
Biomimetic nanoparticles, on the other hand, combine the unique functionalities of natural biomaterials, such as cells or cell membranes, and bioengineering versatility of synthetic nanoparticles, that can be used as an efficient drug delivery platform.
The developed biocompatible exosome-sheathed porous silicon-based nanomedicines for targeted cancer chemotherapy resulted in augmented in vivo anticancer drug enrichment in tumor cells. “This demonstrates the potential of the exosome-biomimetic nanoparticles to act as drug carriers to improve the anticancer drug efficacy”, Santos concludes.
Reference: “Tumor exosome-based nanoparticles are efficient drug carriers for chemotherapy” by Tuying Yong, Xiaoqiong Zhang, Nana Bie, Hongbo Zhang, Xuting Zhang, Fuying Li, Abdul Hakeem, Jun Hu, Lu Gan, Hélder A. Santos and Xiangliang Yang, 23 August 2019, Nature Communications.
DOI: 10.1038/s41467-019-11718-4
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.