Researchers discovered how to flip the structure of complex drug compounds using a simple reagent, offering a game-changing approach for making better medicines.
For the first time, chemists have developed a novel method to manipulate a type of chemical compound that plays a crucial role in many pharmaceuticals, including a medication used to treat breast cancer.
The study, led by researchers at the University of Bristol and published in the journal Nature, also uncovered a surprising reaction mechanism. By introducing a commonly used chemical agent, scientists found they could switch the compound’s configuration from right-handed to left-handed.
Study lead author Varinder Aggarwal, Professor of Synthetic Chemistry at the University of Bristol, said: “The findings change our understanding of the fundamental chemistry of this group of organic molecules. It presents exciting implications because the science allows us to make alternatives of the drug Tamoxifen, with potentially greater potency and less unwanted side effects.”
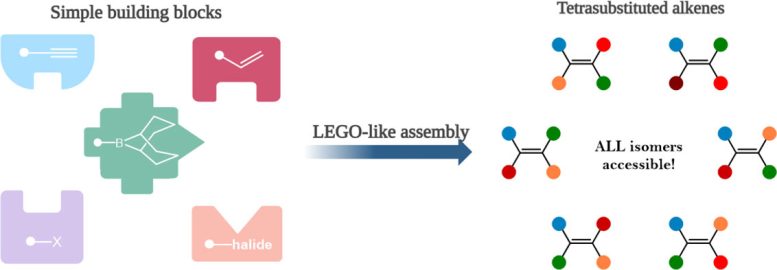
While most alkenes are easy to prepare, a specific type with four different parts – called tetrasubstituted alkenes – are much more challenging but used to make cancer-fighting medicines and natural products like essential oils.
So the research team aimed to find a more efficient method of making tetrasubstituted alkenes, including Tamoxifen, which allows them to be easily manipulated and adapted into different forms.
A Versatile Synthetic Strategy
The new method offers a highly versatile solution to building complex tetrasubstituted alkenes from simple building blocks.
Prof Aggarwal explained: “Our original design plan used organic boronic esters as the key ingredient but that resulted in unstable intermediates, so didn’t work.
“We then tried a less common form of boron-containing molecules, namely boranes and that’s when the clever molecular gymnastics became possible. This new boron system enabled the installation of different groups on the alkene in a controlled manner from very simple building blocks, like Lego.
“It’s so exciting because it holds the key to finding even better drug molecules – like alternatives to Tamoxifen – with more of the properties you want and less of what is undesirable, such as side effects.”
The scientists enlisted the help of computational chemists at Colorado State University to map exactly what was happening. That led to the full extent of their discovery being uncovered.
Applications Beyond Cancer Treatment
Co-author Robert Paton, Professor in Chemistry at Colorado State University, said: “The mechanism showed that by just changing the reaction conditions through adding an agent, the geometry of the alkene can switch direction from left to right. This was surprising and hadn’t been seen before.”
In addition to drug molecules like Tamoxifen, the researchers also worked with natural products such as γ-bisabolene, a fragrant compound found in essential oils, to demonstrate the broad applications of their breakthrough.
Prof Aggarwal added: “Now we have struck upon an effective, flexible methodology, it allows us to swap in other molecules so the potential here is wide-reaching for both drug discovery and materials science.”
Reference: “Boron-mediated modular assembly of tetrasubstituted alkenes” by Liang Wei, Mihai V. Popescu, Adam Noble, Robert S. Paton and Varinder K. Aggarwal, 2 July 2025, Nature.
DOI: 10.1038/s41586-025-09209-2
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
3 Comments
Yes but it’s chiral properties and the chirality of where and how it binds to receptors or elsewhere could make the new version ineffective or toxic or both.
Dr. Kopelson, again you raise astute observations, requiring a sophisticated knowledge of your field to be able to formulate a response.
Stereo selective tamoxifen derivatives have been something of a holy Grail quest for breast cancer researchers for at least two decades.