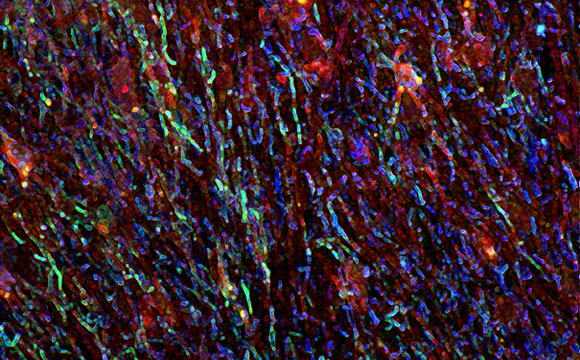
Pictured is immunofluorescence imaging of an active lesion in the brain of an individual with MS.
A newly discovered link between deficits in gut flora and brain inflammation may lead researchers to learn more about its impact on multiple sclerosis.
A team of investigators at Harvard-affiliated Brigham and Women’s Hospital (BWH) has found evidence that suggests that bacteria living in the gut may remotely influence the activity of cells in the brain that are involved in controlling inflammation and neurodegeneration.
“For the first time, we’ve been able to identify that food has some sort of remote control over central nervous system inflammation,” said Francisco Quintana, an investigator in the Ann Romney Center for Neurologic Diseases at BWH and corresponding author on the study. “What we eat influences the ability of bacteria in our gut to produce small molecules, some of which are capable of traveling all the way to the brain. This opens up an area that’s largely been unknown until now: how the gut controls brain inflammation.”
Using pre-clinical models for multiple sclerosis (MS) and samples from MS patients, the team found evidence that changes in diet and gut flora may influence astrocytes in the brain, and, consequently, neurodegeneration, pointing to potential therapeutic targets. The results of their study are published in Nature Medicine.
Previous investigations have suggested a connection between the gut microbiome and brain inflammation, but how the two are linked and how diet and microbial products influence their connection has remained largely unknown. To explore this, Quintana and colleagues performed genome-wide transcriptional analyses on astrocytes — star-shaped cells that reside in the brain and spinal cord — in a mouse model of MS, identifying a molecular pathway involved in inflammation. They found that molecules derived from dietary tryptophan (an amino acid famously found in turkey and other foods) act on this pathway, and that when more of these molecules are present, astrocytes are able to limit brain inflammation. In blood samples from MS patients, the team found decreased levels of the tryptophan-derived molecules.
“Deficits in the gut flora, deficits in the diet or deficits in the ability to uptake these products from the gut flora or transport them from the gut — any of these may lead to deficits that contribute to disease progression,” said Quintana.
The research team plans to investigate this pathway and the role of diet in future studies to determine if the new findings can be translated into targets for therapeutic intervention and biomarkers for diagnosing and detecting the advancement of disease.
This work was supported by grants from the National Institutes of Health, the National Multiple Sclerosis Society, Mallinckrodt Pharmaceuticals, the German Research Foundation, the International Academy of Life Sciences, the Ministry of Science and Technology, Taiwan, and by a fellowship from CAPES, Brazil.
Reference: “Type I interferons and microbial metabolites of tryptophan modulate astrocyte activity and central nervous system inflammation via the aryl hydrocarbon receptor” by Veit Rothhammer, Ivan D Mascanfroni, Lukas Bunse, Maisa C Takenaka, Jessica E Kenison, Lior Mayo, Chun-Cheih Chao, Bonny Patel, Raymond Yan, Manon Blain, Jorge I Alvarez, Hania Kébir, Niroshana Anandasabapathy, Guillermo Izquierdo, Steffen Jung, Nikolaus Obholzer, Nathalie Pochet, Clary B Clish, Marco Prinz, Alexandre Prat, Jack Antel and Francisco J Quintana, 9 May 2016, Nature Medicine.
DOI: 10.1038/nm.4106








“What we eat influences the ability of bacteria in our gut to produce small molecules, some of which are capable of traveling all the way to the brain. This opens up an area that’s largely been unknown until now: how the gut controls brain inflammation.” …Usually the blood brain barrier protects the brain from any onslaught on them by any gut bacteria which goes through the blood. Only meningitis virus can find its route to the brain and also Rabies virus. But virus is just half chemical and half biology. Even if the Bacteria cannot enter the brain through blood (But not virus), the research states that the bacteria is using a CHEMICAL ROUTE, to attack the brain by producing the molecule to gain entry. This aspect of study is worth doing research for development of medicines to curtail its power. Thank You.
Sir i have skin inflammation. What should i do?