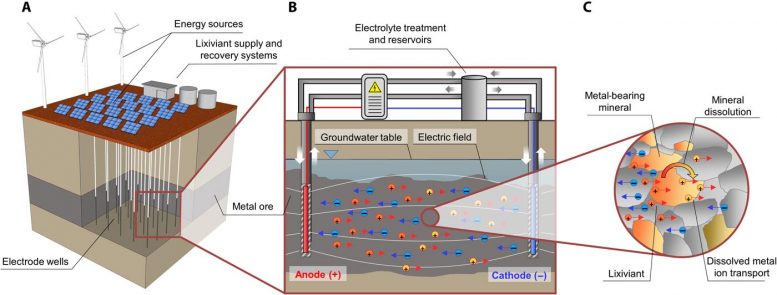
A team of international researchers, including Dr. Rich Crane from the Camborne School of Mines, University of Exeter, have developed a new method to extract metals, such as copper, from their parent ore body.
The research team has provided a proof of concept for the application of an electric field to control the movement of an acid within a low permeability copper-bearing ore deposit to selectively dissolve and recover the metal in situ.
This is in contrast to the conventional approach for the mining of such deposits where the material must be physically excavated, which requires removal of both overburden and any impurities within the ore (known as gangue material).
The researchers believe the new technique has the potential to transform the mining industry, because it has the capability to dissolve metals from a wide range of ore deposits that were previously considered inaccessible.
Furthermore, due to the non-invasive nature of the extraction, the research team are hopeful that the study will help usher in a more sustainable future for the industry.
This is urgently required now in order to provide the plethora of metals required to deliver green technology, such as renewable energy infrastructure and electrified vehicles, whilst limiting any potential environmental damage associated with the mining of such vitally important metals.
The study was recently published in Science Advances.
Electric Fields as a ‘Key-Hole Surgery’ for Mining
Dr. Rich Crane from the Camborne School of Mines, University of Exeter, and co-author of the study, said: “This new approach, analogous to “key-hole surgery”, has the potential to provide a more sustainable future for the mining industry, by enabling the recovery of metals, such as copper, which are urgently needed for our global transition to a new Green Economy, whilst avoiding unwanted environmental disturbance and energy consumption.”
The central principle behind most modern mining techniques has not fundamentally changed since their original conception, which marked the beginning of the Bronze Age: metals are recovered from the subsurface via physical excavation, i.e., the construction of tunnels to gain access to the deposits, or by creating “open cast” mines.
This technique demands large volumes of surface soil, overburden, and gangue material to also be excavated, which can contain millions of tonnes of material – and can also lead to habitat destruction.
In this new publication, experts from the University of Western Australia, the Commonwealth Scientific and Industrial Research Organisation (CSIRO), the Technical University of Denmark, and the University of Exeter, have demonstrated that a targeted electric field can be used to dissolve and then recover copper in situ from the ore – avoiding any requirement to physically excavate the material.
Electromigration: A Novel Mining Mechanism
This new technology comprises the construction (drilling) of electrodes directly into an ore body. An electric current is then applied which can result in the transport of electrically charged metal ions, such as copper, through the rock via a process called electromigration.
The research team have now provided a Proof of Concept for this new technology at a laboratory scale, which has also been verified using computer modeling. They are confident that the idea will work beyond the laboratory scale.
Reference: “Toward a more sustainable mining future with electrokinetic in situ leaching” by Evelien Martens, Henning Prommer, Riccardo Sprocati, Jing Sun, Xianwen Dai, Rich Crane, James Jamieson, Pablo Ortega Tong, Massimo Rolle and Andy Fourie, 30 April 2021, Science Advances.
DOI: 10.1126/sciadv.abf9971
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
14 Comments
An interesting approach. However, what I didn’t see addressed, even in the actual research article, is any concern for post-mining environmental degradation or proposed remediation.
The ‘minining’ will increase the porosity of the host rock, and possibly also the permeability, allowing increased movement of meteoric water. Apparently, the process precipitates jarosite, a common component of acid mine-drainage, and elemental sulfur. This is all occurring below the water table. The FeCl-HCl lixiviant is acidic. Will the sulfur and partially dissolved chalcopyrite oxidize and increase the acidity further? That is, will the abandoned mine(s) become long-lived sources of classic acid mine-drainage?
This is a serious oversight in that much of the public opposition to mining is related to concerns of acid mine-drainage and the surface environmental damage it causes!
And this is published in a “science tech” magazine? First, electricity may melt copper, if you use enough of it, but it cannot “dissolve” copper. Second, you are going to use electrodes miles apart to pass electricity through non-conductive ground? Third, how are you going to generate the tens of thousands of volts and the enormous currents that would be required for this nonsense? Another “computer modeling” fantasy passed off as “science.”
“Electricity … cannot “dissolve” copper.”
I’m afraid you are wrong. See the following:
https://en.wikipedia.org/wiki/Electrolysis
The text of the article doesn’t mention it at all, and the text in the pictures uses odd words, but the process is going to require injecting tons of acid into the ground. It’s going to work like a car battery, and there has to be battery acid in there.
It is going to take a lot of electricity, too. It is like the charging cycle of a car battery, plating metal onto one electrode surface. The energy required to break the metal atoms free from whatever chemical bonds they are in, is considerable, and it’s going to be coming from the acid and the electricity.
All of that is going to be happening underground, out of sight and out of control and confinement.
The process may be better than current practices, overall, but it has some serious issues.
At first look, the main savings is going to be the reduction of the number of people employed.
Looks like they’re using a principle of electrophoresis.
Certainly that’s a valid process if it is working could affect the economics in certain our bodies.
As to the question of environmental damage. It would be unreasonable to expect that in a new mining technique would not have side effects that other mining techniques do not have. It may be, only by operating said mine and observing effects after the fact, will the true environmental effects be observed. However this might be a way to mine deeply more economically than other methods. It would seem to me that deep-mining may be less likely to pollute the surface.
Umm… “control the movement of an acid thru a permeable copper or deposit.”
So were forgoing surface water / runoff contamination for direct groundwater contamination by pumping tens of thousands of gallons of acid directly underground?
Sure, sounds like a great way to permanently ruin freshwater reserves everywhere. At least with surface mining, you can directly control most surface runoff and also any element extraction via solution treatments… while expensive, those processes can be closed and monitored. What these researchers are proposing is am open system where potentially harmful solutions are being pumped underground… that intrinsically cannot be easily controlled or monitored. Try again.
I see a couple people mention filling ground with acid?? I thought the article said a specific acid that is mixed with copper ore would become precipitate during electrolysis application?
It precipitates at the electrodes. However, think about when you wipe down something with a sponge, and then squeeze it to get the water out. Do you get all the water?
Look mom, windmills and solar panels it’s got to be eco friendly for sure!
Bonjour….merci….
So mining has just now realized that fracking is a thing?
So using acid in very large volume is good but open pit mining is bad. What a bunch on morons.
This is the most repetitive article I’ve ever read
what a load of rubbish,they have no idea ,the cost of drilling dead straight holes big enough to put electrodes down will cost ridiculous amounts of dosh,and drilling into hard even for a diamond rig is a slow,keeping the hole dead straight and down a km is impossible,so they think they can pump a shot ton of acid into a watercorse and turn millions of tons of hard rock into a battery ,then suck it out with a straw and she will be alright mate ?? who thought this up obviously hasn’t worked a day in there life on a mine site,what a load of rubbish!!