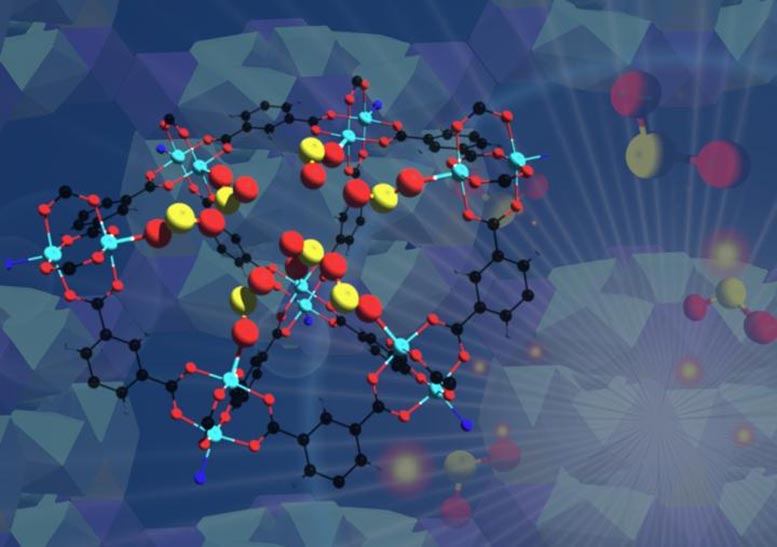
An international team has developed a robust material that can selectively take in toxic sulfur dioxide gas at record concentrations and preserve it for use in chemical production. The researchers verified its performance using a combination of techniques that included X-ray experiments at Lawrence Berkeley National Laboratory’s (Berkeley Lab’s) Advanced Light Source (ALS).
Sulfur dioxide emissions are typically produced by power plants, other industrial facilities, trains, ships, and heavy equipment, and can be harmful to human health and the environment. The team developed porous, cagelike, stable copper-containing molecules known as metal-organic frameworks or MOFs that are designed to separate sulfur dioxide (SO2) gas from other gases. The team exposed the MOF material, dubbed MFM-170, to simulated exhaust gases and found that it efficiently separated out SO2 from the gas mixture at elevated temperatures even in the presence of water.
Existing techniques to remove SO2 from pollution streams can produce a lot of solid and liquid waste and may only remove 60-95 percent of the toxic gas, researchers noted, while the MOF has been shown to eliminate SO2 down to a level below 0.1 parts per million — or 99.99999 percent SO2-free. Their study was published on October 14, 2019, in the journal Nature Materials.
###
Reference: “Reversible coordinative binding and separation of sulfur dioxide in a robust metal–organic framework with open copper sites” by Gemma L. Smith, Jennifer E. Eyley, Xue Han, Xinran Zhang, Jiangnan Li, Nicholas M. Jacques, Harry G. W. Godfrey, Stephen P. Argent, Laura J. McCormick McPherson, Simon J. Teat, Yongqiang Cheng, Mark D. Frogley, Gianfelice Cinque, Sarah J. Day, Chiu C. Tang, Timothy L. Easun, Svemir Rudić, Anibal J. Ramirez-Cuesta, Sihai Yang and Martin Schröder, 14 October 2019, Nature Materials.
DOI: 10.1038/s41563-019-0495-0
The team, led by University of Manchester scientists, used X-rays produced at the ALS to explore the detailed molecular structure of the MOF crystals. They also performed experiments at Oak Ridge National Laboratory; Diamond Light Source, ISIS Neutron, Muon Source, and Manchester University in the U.K.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.