A study suggests that lung epithelial cells act as gatekeepers, guiding the positioning and function of CD4 TRM cells, which play crucial roles in fighting infections, cancers, and asthma. These findings have broad implications for understanding and treating various lung diseases.
Findings have implications in understanding, preventing and treating multiple lung diseases.
Lung immunity is essential to combat all pulmonary diseases, including COVID-19, pneumonia, lung cancer, asthma, and COPD. Lung immunity differs from the systemic immunity which is the normal focus of biomedical investigations and interventions, but factors influencing the establishment and regulation of lung immunity are mostly still unknown. Now a new study reveals lung cell roles in guiding the immune system.
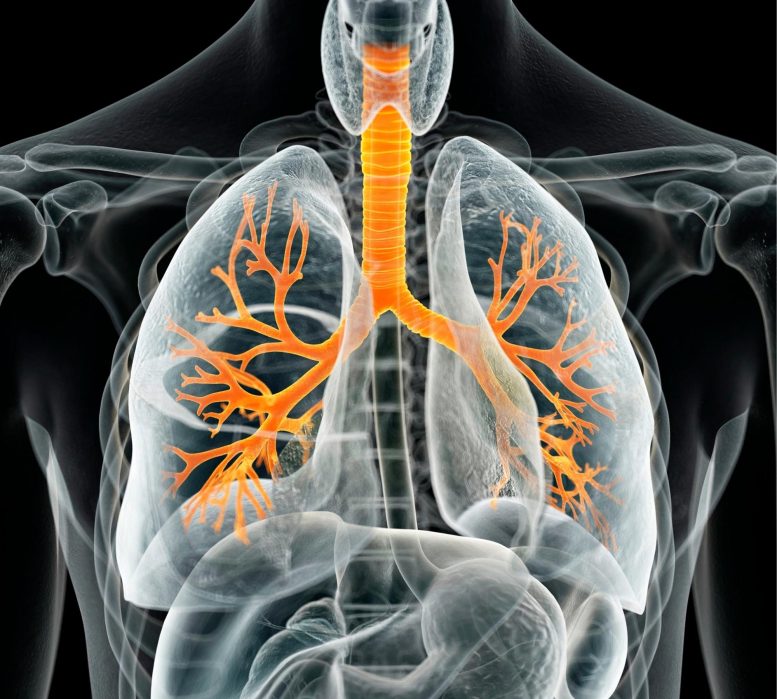
Researchers from Boston University School of Medicine (BUSM) have found that control of immunity in the lungs is accomplished by cells that line the air spaces, epithelial cells, using a specialized immune-facing molecule, MHC-II. This epithelial MHC-II is essential for localizing and scripting highly specialized immune cells called resident memory T (TRM) lymphocytes inside the lungs.
“Epithelial cells in the lung are typically envisioned as supporting respiratory functions, while MHC-II is understood to connect immune cells to immune cells, so the finding that MHC-II on lung epithelial cells tells TRM cells where to go and what to do in the lung is novel and unanticipated,” explained corresponding author Joseph Mizgerd, ScD, professor of medicine, microbiology, and biochemistry at BUSM.
By analyzing the lung epithelial cells from human and experimental models, the researchers learned that all the different types of epithelial cells examined express MHC-II and increase its expression during infections. The only known function of MHC-II is to educate immune cells called CD4+ T cells. In cell cultures, lung epithelial cells could use this molecule to tell T cells what to do, so that they would be able to respond appropriately to microbes that might cause an infection. Interrupting only MHC-II only on lung epithelial cells led to aberrant numbers and types and places of CD4+ T cells in the lungs, but not the blood, revealing these specific lung cells were responsible for directing pulmonary immunity.
“Our study suggests that lung epithelial cells are akin to gatekeepers that are tasked with appropriately instructing locations of CD4 TRM cell outposts and their ability to fight future infections. Given that TRM cells, beyond their protective roles in pneumonia, play key roles in fighting cancers and provoking asthma, our findings have larger implications in understanding, preventing, and treating multiple lung diseases,” said first author Anukul Shenoy, PhD, postdoctoral scientist in BUSM’s Pulmonary Center.
In addition to showing that lung epithelial cells use MHC-II to organize the immune system in the lung, the study also reveals two unexpected findings that grew out of the main discovery. First, other immune-facing molecules depend on MHC-II for getting to the cell surface where they can interact with other cells to do their immune instruction. Second, that the lack of MHC-II on lung epithelial cells causes changes to the local lung immune system that mirror a rare but serious outcome of immune-targeting cancer therapies (“checkpoint inhibitor therapies”). “This led to the discovery that a molecular target of these cancer treatments is one of the molecules that depends on MHC-II for getting to the cell surface, and the suggestion that deleterious side effects from checkpoint inhibitor therapies may result from inhibition of immune cell guidance by lung epithelial cells,” said Mizgerd.
The researchers envision that interventions (both preventative and remedial) can be designed to harness the ability of lung epithelial cells in modulating the pulmonary immunity. “That way we would be able to use the patients’ own lung epithelial cells to turn on protective roles of TRM cells during pneumonia and/or cancers, while being able to shut off their pathological roles during asthma, as deemed necessary,” said Shenoy.
These findings appear online in the journal Nature Communications.
Reference: “Antigen presentation by lung epithelial cells directs CD4+ TRM cell function and regulates barrier immunity” by Anukul T. Shenoy, Carolina Lyon De Ana, Emad I. Arafa, Isabelle Salwig, Kimberly A. Barker, Filiz T. Korkmaz, Aditya Ramanujan, Neelou S. Etesami, Alicia M. Soucy, Ian M. C. Martin, Brian R. Tilton, Anne Hinds, Wesley N. Goltry, Hasmeena Kathuria, Thomas Braun, Matthew R. Jones, Lee J. Quinton, Anna C. Belkina and Joseph P. Mizgerd, 5 October 2021, Nature Communications.
DOI: 10.1038/s41467-021-26045-w
Funding for this study was provided by NIH grants including HL147397 to EIA, HL142199 to KAB, HL147461 to FTK, HL136725 to MRJ, GM120060 and HL111449 to LJQ, AI115053, HL135756, and HL137081 to JPM and T32 HL007035 for support of trainees in addition to support by the German Research Foundation (DFG) Clinical Research Group KFO309 TP08, the Excellence Cluster Cardio-Pulmonary Institute (CPI), and the German Center for Lung Research (DLZ) to TB.







I could have bet my life that my husband would never recover from cancer, I never imagined he could beat this disease lung cancer because we lost hope when chemo and radiation which was the only alternative suggested to us didnt help. After he got diagnosed with lung cancer the doctor told us that he can’t be cured and we will leave everything about his health in Allah’s hands but can take some medications and have some chemotherapy to shrink the growths and despite all they kept coming back after some years. We tried every possible remedy we could both natural, therapies and multivitamins but there was no solution, weeks went by and then months and he kept on getting worse day by day. He could barely talk, eat or move his limbs, he lost abilities completely! .. Though everyone lost hope I never gave up on him until fate directed us to Dr Jude herbal home, a herb doctor from who cures all cancer and tumors with herbal formula (pentosin). We made our order after contacting him through his email drjudeherbalhome@gmail .com and InshaAllah my husband won the fight against this disease completely with all complications reversed. Check out his page Dr Jude Herbal Home for info, I am so grateful for bringing Dr Jude and nature to this planet