A team of scientists from the Department of Neurogenetics at the Max Planck Institute and University Medical Center Göttingen has discovered a new treatment approach for the hereditary neurological disorder Charcot-Marie-Tooth disease type 1A.
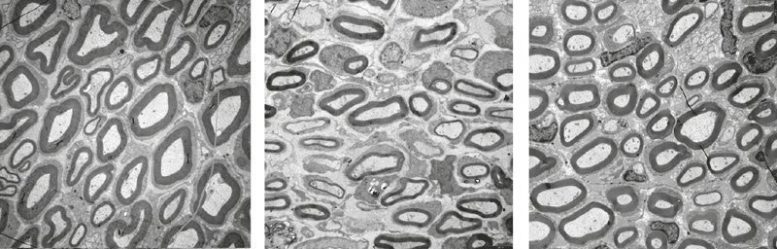
Charcot-Marie-Tooth disease type 1A is the most common inherited disease affecting the peripheral nervous system. Researchers from the Department of Neurogenetics at the Max Planck Institute of Experimental Medicine and University Medical Center Göttingen have discovered that the maturity of Schwann cells is impaired in rats with the disease. These cells enwrap the nerve fibers with an insulating layer known as myelin, which facilitates the rapid transfer of electrical impulses. If Schwann cells cannot mature correctly, an insufficient number of nerve fibers is covered with myelin during development. According to the researchers, the growth factor neuregulin-1 has immense therapeutic potential: rats treated with neuregulin-1 have more myelinated nerve fibers. The symptoms of the disease diminish significantly as a result.
Patients with Charcot-Marie-Tooth disease type 1A harbor an extra copy of the PMP22 gene which leads to the overproduction of the peripheral myelin protein 22 (PMP22), a key component of myelin. This causes slow, progressive nerve damage, which can begin as early as childhood. Patients suffer from numbness, tingling, and pains in the arms and legs, as well as weakness of leg and arm muscles. Some patients can only move around with the help of a wheelchair. The disease has been incurable to date, as little is known about the fundamental mechanisms of the disorder.
The researchers in Göttingen have now studied genetically modified rats, which, like Charcot-Marie-Tooth patients, produce too much PMP22. In these animals, the Schwann cells cannot mature properly, with the result that insufficient numbers of axons are enwrapped with myelin during development. “This is caused by an imbalance of two signaling pathways in the Schwann cells – the PI3K/AKT and the MEK/ERK signaling pathway – which are important for cell maturation,” explains Robert Fledrich from the Max Planck Institute of Experimental Medicine.
In this latest study, the scientists tested the therapeutic potential of the growth factor neuregulin-1. They were able to show that the balance between the two signaling pathways could be restored by administering neuregulin-1. The growth factor is normally produced by nerve cells and acts as an important maturation signal for Schwann cells during development. In the case of acute nerve damage, it is also formed by Schwann cells and plays an important role in repairing damaged nerves. “In genetically modified rats, even a brief neuregulin-1 treatment within the first two weeks of life improves the animals’ disease symptoms until they reach adulthood,” says co-author Ruth Stassart.
The scientists and the neurologists from Michael Sereda’s Research Group now want to conduct additional studies to drive forward the development of a treatment for Charcot-Marie-Tooth disease type 1A. Applying the findings to patients, however, is still a long way off. A therapy with Neuregulin itself is not safe for patients. Therefore, drugs that can imitate the neuregulin-1 signaling pathway will be tested. The recently established Germany-wide network for Charcot-Marie-Tooth disease (CMT-NET) should help in this regard. Patients, researchers, and doctors will be able to use the network in Germany to find out about the progress being made in researching this little-known disease.
Reference: “Soluble neuregulin-1 modulates disease pathogenesis in rodent models of Charcot-Marie-Tooth disease 1A” by Robert Fledrich, Ruth M Stassart, Axel Klink, Lennart M Rasch, Thomas Prukop, Lauren Haag, Dirk Czesnik, Theresa Kungl, Tamer A M Abdelaal, Naureen Keric, Christine Stadelmann, Wolfgang Brück, Klaus-Armin Nave and Michael W Sereda, 24 August 2014, Nature Medicine.
DOI: 10.1038/nm.3664
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.