
UT Southwestern discovery could lead to a novel approach for treating heart disease and fatty liver disease.
Two researchers at UT Southwestern Medical Center have discovered a protein that helps regulate how the liver releases cholesterol-carrying particles into the bloodstream. The finding could eventually support the development of new therapies for both atherosclerotic heart disease and fatty liver disease.
The research, reported in the American Heart Association journal Circulation, identified a protein called HELZ2 as an important regulator of apolipoprotein B (APOB). This gene is essential for producing apoB proteins, which are necessary for assembling lipoproteins, the particles that move cholesterol and fats through the bloodstream.
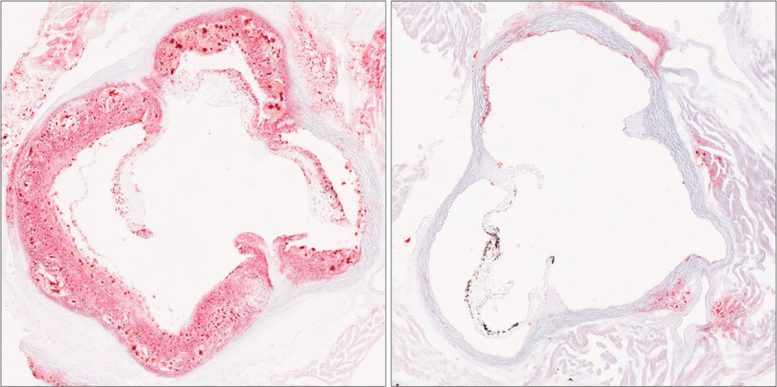
“These particles are a major driver of plaque buildup in the arteries,” said senior author Zhao Zhang, Ph.D., Assistant Professor in UT Southwestern’s Center for the Genetics of Host Defense and of Internal Medicine. “What we found is that HELZ2 acts as a powerful control point for how many cholesterol-carrying particles ultimately enter the bloodstream.”
The team discovered that HELZ2 influences the stability of APOB messenger RNA (mRNA), the molecule that carries genetic instructions from DNA to the cellular machinery that builds proteins. Inside liver cells, HELZ2 reduces how long APOB mRNA persists. When the protein is more active, the APOB message breaks down more quickly. This leads to lower production of apoB protein and fewer cholesterol-carrying particles released into circulation.
“Most previous research focused on what happens to apoB after it’s already made,” said Yiao Jiang, Ph.D., a postdoctoral researcher in the Zhang Lab and study co-author. “What surprised us is that HELZ2 acts much earlier, by controlling how long the apoB ‘message’ survives before the protein is even produced.”
Scientists have long studied how apoB proteins are processed after they form, but this discovery points to an earlier step in the process. By controlling how long the genetic instructions remain available inside liver cells, HELZ2 effectively determines how many lipoproteins can be produced in the first place.
A genetic mutation reveals a cholesterol–liver fat tradeoff
To uncover this mechanism, the researchers relied on a large-scale genetic screening approach originally developed by Nobel Laureate Bruce Beutler, M.D., Director of the Center for the Genetics of Host Defense and Professor of Immunology and Internal Medicine at UT Southwestern. The team examined mice with unusual patterns of liver fat accumulation and traced the effect to a mutation that increased HELZ2 activity.
This gain-of-function mutation made HELZ2 more active and reduced the stability of APOB mRNA in liver cells.
Mice carrying the mutation produced fewer circulating lipoproteins, including LDL (low-density lipoprotein) cholesterol and triglycerides. Lower levels of these particles reduced their risk of developing atherosclerosis. However, the same animals accumulated more fat in their livers, revealing an important biological tradeoff between cholesterol levels in the blood and fat storage in liver tissue. Mice without the mutation showed the opposite pattern, with higher blood lipids but less fat buildup in the liver.

“We can think of HELZ2 as a kind of dial between the liver and the bloodstream,” Dr. Zhang said. “Turning it up lowers cholesterol in the blood but increases liver fat. Turning it down does the reverse. That balance makes HELZ2 especially interesting as a potential therapeutic target.”
The finding highlights how tightly the liver balances two critical processes: exporting fats into the bloodstream and storing them internally. Changing HELZ2 activity appears to shift that balance.
Targeting HELZ2 could open new treatment strategies
Today, statins remain the most widely prescribed medications for lowering cholesterol and reducing the risk of heart disease. The researchers say that further work could reveal whether HELZ2 can be targeted to reduce harmful lipoproteins through a different biological pathway.
If scientists learn how to fine-tune HELZ2 activity, it could lead to treatments designed to adjust the liver’s production of cholesterol-carrying particles. That same approach might also provide new strategies for managing fatty liver disease, which affects millions of people worldwide.
“The idea that we can control apoB at the RNA level represents a major shift in how we think about cholesterol regulation,” Dr. Zhang said. “It gives us a new molecular lever – and potentially a new set of tools – for tackling these conditions.”
Reference: “HELZ2 Regulates Apob mRNA Stability to Modulate Fatty Liver Disease and Atherosclerosis” by Yiao Jiang and Zhao Zhang, 25 December 2025, Circulation.
DOI: 10.1161/CIRCULATIONAHA.125.076468
This research was supported by funding from the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health (R00DK115766 and R01DK130959).
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
8 Comments
Gwen Tennyson
All you need and its pretty affordable, is Retatrutide, Mots-C and BPC-157 and your liver will be clean and renewed within 30-40days! 100% healed.
Big pharmacy may stop it.
Go to hell!
All you need and its pretty affordable, is Retatrutide, Mots-C and BPC-157 and your liver will be clean and renewed within 30-40days! 100% healed.
This is fantastic! Hoping this will be available to the public soon with minimal side effects. Thank you so much to the researchers and NIH.
very interested in this research my family has a history of Familial Hypercholesterolemia this looks like it might be a great help.
What is the pill called do they have people that have cirrhosis