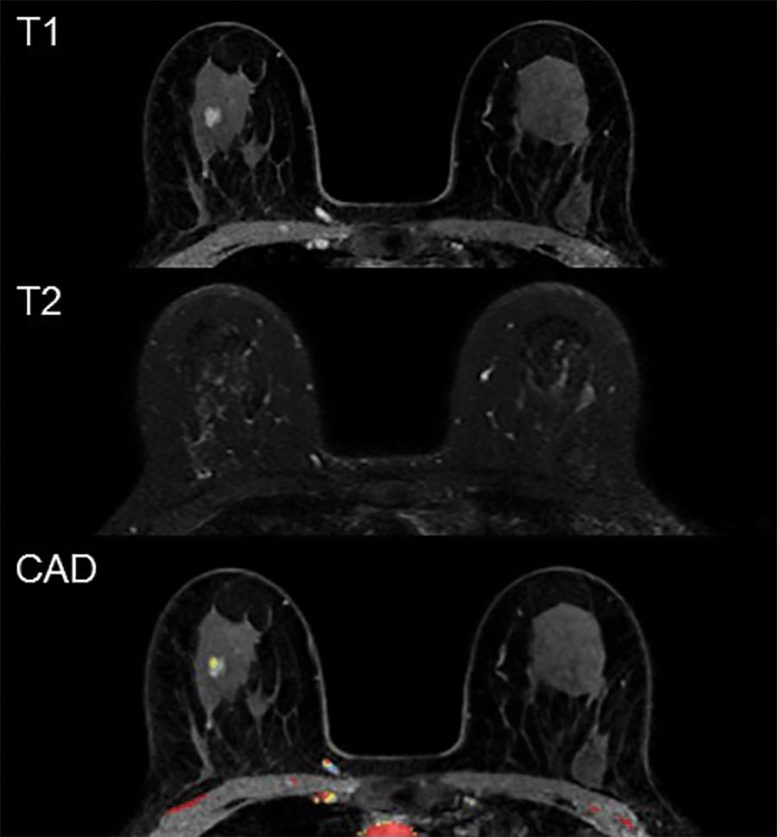
Example of a false-positive MRI. In a 59-year-old participant, first-round screening MRI showed an 8-mm irregular mass in the right breast. The lesion shows rapid heterogeneous type 2 enhancement, low T2 signal intensity, and no evident diffusion restriction. Histopathologic findings showed sclerotic stroma and apocrine metaplasia. Credit: Radiological Society of North America
Prediction models based on clinical characteristics and imaging findings may help reduce the false-positive rate in women with dense breasts who undergo supplemental breast cancer screening with MRI, according to a new study in the journal Radiology.
Women with dense breast tissue have a much higher risk of breast cancer compared to those with average breast density. High breast density also markedly reduces the sensitivity of mammography due to the masking effect of the fibroglandular tissue, meaning that cancers can be hidden within dense breast tissue.
For these reasons, breast MRI is considered a potentially useful supplement to mammography screening in women with dense breast tissue. It is the most sensitive imaging technique for diagnosing breast cancer and can differentiate well between lesions and abnormalities of the breast. Research has confirmed its substantial added value as a screening tool for women at high risk of breast cancer.
However, the high sensitivity that makes MRI an excellent screening tool also means it often detects benign lesions that otherwise would have gone unnoticed. Women who get recalled for additional work-up based on these findings potentially face repeat MRI scans, targeted ultrasound, and biopsy. The need for additional investigations may cause anxiety in the patient, increase health care costs or lead to biopsy-related complications.
“The reduction of the false-positive recall rate is an important issue when considering the use of breast MRI as a screening tool,” said study lead author Bianca M. den Dekker, M.D., from the University Medical Center Utrecht in Utrecht, the Netherlands.
In the new study, Dr. den Dekker and colleagues developed prediction models to distinguish true-positive MRI screening from false-positives. To create the models, they combined MRI findings with clinical characteristics like body mass index, family history of breast cancer and use of hormone replacement therapy.
They used data from the Dense Tissue and Early Breast Neoplasm Screening (DENSE) trial, which evaluated the effectiveness of screening with mammography plus MRI compared to mammography alone in Dutch breast cancer screening participants aged 50 to 75 years, with extremely dense breasts.
Of the 454 women who had a positive MRI result in a first supplemental MRI screening round, 79 were diagnosed with breast cancer, meaning that 375 women had false-positive MRI examinations. The full prediction model, based on all collected clinical characteristics and MRI findings, could have prevented 45.5% of false-positive recalls and 21.3% of benign biopsies, without missing any cancers. The model solely based on readily available MRI findings and age had a comparable performance and could have prevented 35.5% of false-positive MRI screenings and 13.0% of benign biopsies.
“Our prediction models may identify a substantial number of false-positives after first-round supplemental MRI screenings, reducing false-positive recalls and benign biopsies without missing any cancers,” Dr. den Dekker said. “This brings supplemental screening MRI for women with dense breasts one step closer to implementation.”
The researchers intend to perform validation studies using data from different populations. They also want to study the performance of prediction models in subsequent screening rounds. Dr. den Dekker noted that the false-positive rate in the study group fell from 79.8 per 1,000 screenings in the first round to 26.3 per 1,000 in the second.
“This can be partly explained by the availability of prior MRI examinations, which allows comparison for interval change,” she said. “As incident screening rounds have a much lower false-positive rate, separate models may have to be created.”
Reference: “Reducing False-Positive Screening MRI Rate in Women with Extremely Dense Breasts Using Prediction Models Based on Data from the DENSE Trial” by Bianca M. den Dekker, Marije F. Bakker, Stéphanie V. de Lange, Wouter B. Veldhuis, Paul J. van Diest, Katya M. Duvivier, Marc B. I. Lobbes, Claudette E. Loo, Ritse M. Mann, Evelyn M. Monninkhof, Jeroen Veltman, Ruud M. Pijnappel and Carla H. van Gils , For the DENSE Trial Study Group, 17 August 2021, Radiology.
DOI: 10.1148/radiol.2021210325







Be the first to comment on "Reducing False-Positives in MRI Breast Cancer Screening"