Fusion energy, using deuterium-tritium fuel, offers a potential for limitless and clean energy.
Fusion energy has the potential to supply safe, clean, and nearly limitless power. Although fusion reactions can occur for light nuclei weighing less than iron, most elements will not fuse unless they are subject to immense temperatures and pressures, such as in the interior of a star. To create burning plasmas in experimental fusion power reactors such as tokamaks and stellarators, scientists seek a fuel that is relatively easy to produce, store, and bring to fusion. The current best bet for fusion reactors is deuterium-tritium fuel. This fuel reaches fusion conditions at lower temperatures compared to other elements and releases more energy than other fusion reactions.
Deuterium and tritium are isotopes of hydrogen, the most abundant element in the universe. Whereas all isotopes of hydrogen have one proton, deuterium also has one neutron, and tritium has two neutrons, so their ion masses are heavier than protium, the isotope of hydrogen with no neutrons. When deuterium and tritium fuse, they create a helium nucleus, which has two protons and two neutrons. The reaction releases an energetic neutron. Fusion power plants would convert the energy released from fusion reactions into electricity to power our homes, businesses, and other needs.
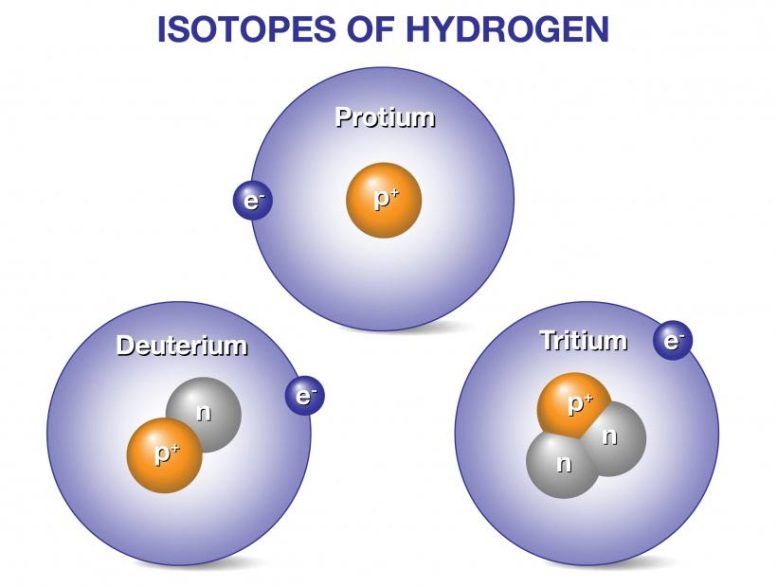
Fortunately, deuterium is common. About 1 out of every 5,000 hydrogen atoms in seawater is in the form of deuterium. This means our oceans contain many tons of deuterium. When fusion power becomes a reality, just one gallon (3.8 liters) of seawater could produce as much energy as 300 gallons (1,135 liters) of gasoline.
Tritium is a radioactive isotope that decays relatively quickly (it has a 12-year half-life) and is rare in nature. Fortunately, exposing the more abundant element of lithium to energetic neutrons can generate tritium. A working fusion power plant would need enriched lithium to breed the tritium it needs to close the deuterium-tritium fuel cycle. Current R&D efforts are focused on advanced designs of tritium breeding blankets using lithium originally obtained from Earth-based sources.
Deuterium-Tritium Fuel Facts
- Water made from deuterium is about 10 percent heavier than ordinary water. That’s why it is sometimes referred to as “heavy water.” It will actually sink to the bottom of a glass of ordinary water.
- Sources of tritium on Earth include natural production from interactions with cosmic rays, energy-producing nuclear fission reactors such as the heavy water CANDU reactor, and nuclear weapons testing.
- To avoid certain R&D challenges including structural material damage from energetic neutrons, fusion scientists are interested also in aneutronic fusion reactions (such as deuterium-helium-3 and proton-boron fusion) even though these fusion reactions occur at higher ion temperatures than for deuterium and tritium.
DOE Office of Science: Contributions to Deuterium-Tritium Fuel
Part of the mission of The Department of Energy Office of Science, Fusion Energy Sciences (FES) program is to develop a practical fusion energy source. FES works with the Advanced Scientific Computing Research program using scientific computing to advance fusion science and understand the effect of ion mass on various plasma phenomena. At Office of Science user facilities such as the DIII-D tokamak and NSTX-U spherical tokamak, scientists study the impact of ion mass on plasma confinement, transport, and turbulence. The confinement of fusion products such as the helium ion is also studied in presence of helical magnetic fields. The Office of Science Nuclear Physics program develops the fundamental nuclear science underpinning the understanding of fusion by creating nuclear reaction databases, generating nuclear isotopes, and elucidating aspects of nucleosynthesis.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
6 Comments
I want a fusion reactor the size and cost of a microwave oven that can power my home, car and spaceship. Gimme, gimme…
“Although fusion reactions can occur for light nuclei weighting less than iron”
Should that not read “less than Cobalt”?
From the web: Of all nuclides, iron-56 has the lowest mass per nucleon. With 8.8 MeV binding energy per nucleon, iron-56 is one of the most tightly bound nuclei
When we get a working fusion reactor, I hope you get bonuses in United States ” created ” gold coins. Thank you Department of Energy Office of Science, Fusion Energy Sciences for your fusion energy work. ” see Nuclear transmutation Wikipediahttps://en.wikipedia.org
Nuclear transmutation
” Under true nuclear transmutation, it is far easier to turn gold into lead than the reverse reaction, which was the one the alchemists had ardently pursued.”
Are you guys serious??? How long are these scientists gonna keep playing with fantasy??? All of these scientists are wasting, Yes, wasting their lifetimes, and our disaster-warming planet on creating fusion conditions for tiny bits of un-obtainable fuel. Currently, for the next hundred years, fusion energy production and harvesting is not possible nor economically feasible. YOU MUST REALIZE THAT THIS WILL NEVER PRODUCE ANY ELECTRICITY FOR ANY OF OUR GREAT-GRANDCHILDRENS’ CHILDREN. WHO IS GOING TO SPEAK UP??? We can only use what hyper-novas have given us, Uranium and Thoreum. Uranium232 (from Thoreum) is not Uranium235. We have used the WORST nuclear process to generate energy. We have used the WORST nuclear process to generate electricity and two-hundred thousand year wastes. Politicians are the stupidest scientists. THE ONLY ANSWER to OUR PLANTETARY-SIZED problem is looking at OUR STUPID HUMAN SOCIETY, IN PITY. The only way to possibly catch up to a hot, dying planet, is to fully utilize Thoreum (converts to Uranium232; aka. the unmatched and incredible-nuclear fuel) … To Immediately Convert almost all nuclear plants, as well as all retired nuclear or non-nuclear electrical facilities for power and high (600F) industrial heat using Uranium232-modules. The Uranium232 is ideal for modular power, at any location on Earth. The Uranium232 ‘salt’ form is a liquid at normal atmospheric pressure. It’s temperature rises to over 600F, but not more than that. If it cools down, it solidifies. Aahhhh, I need sleep…. Wake-up world.
Whoops, … In my last comment, I was estimating the upper-temperature for the Uranium232 liquid-florine salt to be above 600F. My memory had failed me….. The upper-temperature range for the molten-salt, UF6, super-dooper-fuel is around 700C. That is quite a difference and this very high temperature is an example of how incredibly advantageous Thoreum-based nuclear energy is in comparison to Uranium235’s numerous costly short-falls.