Scientists Have Identified New Cancer Treatment Targets
In Switzerland, cancer is the second-leading cause of death. Non-small cell lung cancer (NSCLC) is the cancer form that kills the most people and is still mostly incurable. Unfortunately, only a small percentage of patients survive the metastatic stage for a long time, and even recently approved therapies can only prolong patients’ lives by a few months. As a result, researchers are looking for innovative cancer treatments. Researchers from the University of Bern and the Insel Hospital identified new targets for drug development for this cancer type in a recent study published in the journal Cell Genomics.
The Dark Matter of the Genome
They searched for novel targets in the poorly understood class of genes known as “long noncoding RNAs (Ribonucleic acids)” (lncRNAs). LncRNAs are abundant in the “Dark Matter,” or non-protein-coding DNA, which makes up the overwhelming bulk of the human genome. The human genome comprises around 20’000 “classical” protein-coding genes, but they are dwarfed by 100’000 lncRNAs. The biological functions of 99% of lncRNAs remain unknown.
As the name long noncoding RNAs implies, unlike messenger RNAs (mRNAs), they do not encode the construction plans for proteins. Like for mRNAs, the building instructions for lncRNAs are contained in the cell’s DNA.
A New Tool Determines Potential Targets
To study the role of lncRNAs in NSCLC, the researchers started by analyzing publicly available datasets to see which lncRNAs are present in NSCLC. This analysis led to a list of over 800 lnRNAs, whose importance for NSCLC cells the researchers wished to investigate. For this investigation, they developed a screening system that prevents the production of the selected lncRNAs by deleting part of their construction instructions in the DNA.

They applied their screening system to two NSCLC cell lines derived from patients and looked at how the inhibition of the selected lncRNAs affected so-called “hallmarks” of cancer cells. Hallmarks are cellular behaviors that contribute to disease progression: Proliferation, metastasis formation, and therapy resistance. “The advantage of assessing three different cancer hallmarks is that we have a comprehensive view but also have substantial amounts of data from different experiments, from which we needed to derive a single list of long noncoding RNAs that are important for non-small cell lung cancer,” says Rory Johnson, Assistant Professor at the University of Bern, who led the NCCR RNA & Disease funded project. The analysis yielded in the end a list of 80 high-confidence candidate lncRNAs important for NSCLC out of the over 800 investigated. From these 80, the researchers picked out several lncRNAs for follow-up experiments.
Destroying a Long RNA With a Short One
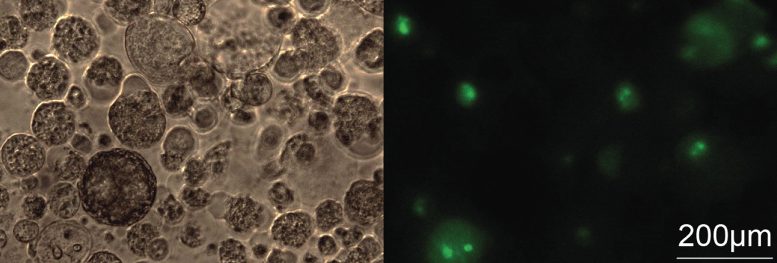
For these follow-up experiments, an approach was used, which does not work at the DNA level but targets lncRNAs after their production. For this purpose, the researchers used small chemically-synthesized RNAs called Antisense Oligonucleotides (ASOs), which bind to the lncRNAs they target and lead to their degradation. Of note, several ASOs are approved for treating human diseases, although none yet for cancer.
These follow-up experiments showed that, for the majority of the picked lnRNAs, their destruction by an ASO inhibited cancer cell division in cell culture. Importantly, the same treatment produced little if any effect on non-cancerous lung cells, which should not be harmed by the cancer treatment. In a 3-dimensional model of NSCLC, which more closely resembles the tumor than cell culture, the inhibition of a single lncRNA with an ASO reduced the tumor growth by more than half. “We were positively surprised to see how well the antisense oligonucleotides could restrain tumor growth in different models,” states Taisia Polidori, co-first author, who worked on the project as part of her doctoral thesis research at the University of Bern.
Therapy Development and Application to Other Tumor Types
The researchers are continuing their research in pre-clinical cancer models and are considering collaborating with existing companies or creating a startup in order to develop a drug to treat patients. Regarding other cancers, Roberta Esposito, co-first author and postdoc at the University of Bern: “Like a telescope that can be quite easily repositioned to study a different part of space, our approach should be easily adaptable to reveal new potential treatment types for other cancer types.” Dr. Esposito will now apply the “telescope” to identify new targets for colorectal cancer. For this purpose, she has received funding from the Medical Faculty of the University of Bern donated by the Béactrice Ederer-Weber foundation.
NCCR RNA & Disease – The role of RNA in Disease Mechanisms
The NCCR RNA & Disease – The Role of RNA Biology in Disease Mechanisms studies one very important class of molecules of life: RNA (ribonucleic acid), which is pivotal for many vital processes and is functionally much more complex than initially assumed. For instance, RNA defines the conditions, in a given cell, under which a given gene is or is not activated. If any part of this process of genetic regulation breaks down or does not run smoothly, this can cause heart disease, cancer, brain disease, and metabolic disorders. The NCCR brings together Swiss research groups studying different aspects of RNA biology. By researching which regulatory mechanisms are dysregulated in disease, the NCCR discovers new therapeutical targets.
Reference: “Multi-hallmark long noncoding RNA maps reveal non-small cell lung cancer vulnerabilities” by Roberta Esposito, Taisia Polidori, Dominik F. Meise, Carlos Pulido-Quetglas, Panagiotis Chouvardas, Stefan Forster, Paulina Schaerer, Andrea Kobel, Juliette Schlatter, Erik Kerkhof, Michaela Roemmele, Emily S. Rice, Lina Zhu, Andrés Lanzós, Hugo A. Guillen-Ramirez, Giulia Basile, Irene Carrozzo, Adrienne Vancura, and Rory Johnson, 22 August 2022, Cell Genomics.
DOI: 10.1016/j.xgen.2022.100171
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Interesting article. Only concern is that lncRNA is “pootlrly understood” and when it is, they may say “cr@p! Shouldn’t’ve touched that! Did more damage than good…”