Light controlled current transport by charged atoms, now demonstrated for the first time, makes new applications conceivable.
Light makes some materials conductive in a previously unforeseen way. In common silicon solar cells, electrons flow when the sun shines. However, scientists at the Stuttgart-based Max Planck Institute for Solid State Research have now come up with a surprise: in a special perovskite, another material used for solar cells, light not only releases electrons, but also electrically charged atoms, known as ions. Moreover, this novel photoeffect is extremely large. Ion conductivity increased by a factor of one hundred. For solar cells made from the material investigated here, the high light-induced ion conductivity is rather damaging; the consequences, however, can now be specifically counteracted. From the point of view of the researchers in Stuttgart, the effect is ground-breaking in itself, as it makes novel, light-controlled electrochemical applications conceivable, such as batteries directly charged by light.
When it comes to efficiency, silicon solar cells set standards. But especially for photovoltaic elements with particularly high efficiencies, silicon production is both complex and expensive. Materials referred to as perovskites, owing to their structure, could offer a less expensive alternative here. A team of scientists headed by Joachim Maier, Director at the Max Planck Institute for Solid State Research, have now examined how light influences the transport of electricity in these materials based on the perovskite methylammonium lead iodide (MAPI). Their interest in these materials was aroused during a collaboration with Michael Grätzel, who conducts research at the École polytechnique fédérale de Lausanne (EPFL) and is an External Scientific Member of the Stuttgart-based Max Planck Institute.
In their experiments, the researchers now observed that ions, that is charged atoms, contribute to conductivity to an unexpectedly high degree when the material is illuminated. In perovskite solar cells, the effect can lead to structural alterations and impair efficiency. “However, our findings may help to prevent such aging processes,” says Joachim Maier. For the chemist, however, the phenomenon as such is most exciting primarily because it creates the fundamental possibility of releasing mobile ions with the help of light, namely those charge carriers which transport electricity in electrochemical applications such as batteries, fuel cells, or electrochemical sensors and switches.
A mechanism translates the holes generated by light into ion conductivity
That light influences ion transport has only previously been demonstrated in biology: Illumination is able to indirectly alter the permeability of a cell membrane. “Very surprising, however, is the fact that the ionic conduction of crystalline solids can be directly modified and to what extent this is possible,” says Joachim Maier. His team observed how the number of free iodide ions was increased by a factor of one hundred. Ionic conduction is thus increased to a similar extent as that known for light-induced electronic conductivity.
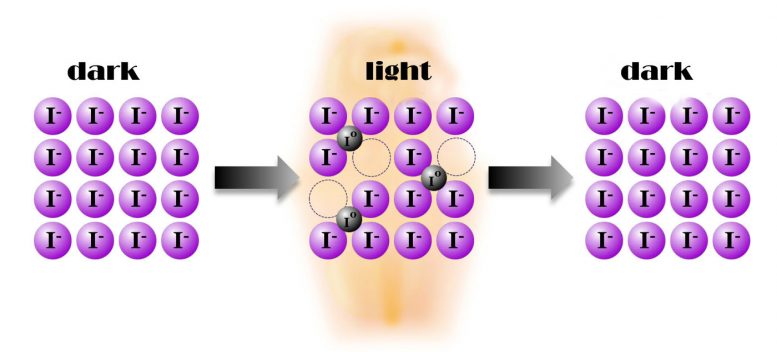
The researchers in Stuttgart not only demonstrated the phenomenon experimentally. They can also explain it. According to them, the light initially releases electrons, as is usual in solar cells. The negatively charged electrons leave positively charged holes in the crystal lattice, as physicists would say. These neutralize otherwise negatively charged iodide ions within the crystal. Because an uncharged iodine atom is much smaller than an iodide ion, it occupies a so-called interstitial space, that is, a free space in the crystal lattice in which the larger iodide ion does not fit. The resulting gaps in the crystal lattice allow ion conduction in much the same way as electron holes allow electron conduction. “Crucial to this effect is that there’s a mechanism that translates the holes created by light directly into ionic conductivity,” says Joachim Maier.
Various methods prove the effect beyond any doubt
The researchers used various methods to prove the effect beyond any doubt. In an initial experiment, they used electrical contacts for MAPI that blocked ions, that is, they allowed only the electrons to pass. They used a specific current and measured the voltage. If ions are involved in the current flow, the voltage should increase after a short time, because they can only move initially, but are then blocked by the contacts. This was exactly what the researchers in Stuttgart observed.
Clear evidence of ionic conduction was also provided by the voltage measured in an open circuit, which was generated by the researchers using the perovskite as the electrolyte phase of an illuminated battery cell: if electrons in the material primarily transported current, a short circuit would occur, and no voltage would be produced. However, using an ionically conductive material as the electrolyte, the anticipated battery voltage can be measured.
The researchers directly demonstrated iodine transport in two further experiments. They exposed one side of the perovskite to gaseous iodine. They attached a copper film to the other side, which acts as a so-called iodine sink because of its propensity to react to form copper iodide. Under illumination, this process occurred at very high speed. Transport of the iodine within the perovskite sample was also demonstrated by an experiment in which toluene acted as an outer sink for the element. The researchers demonstrated spectroscopically that the iodine concentration in the toluene increased as soon as the perovskite was illuminated.
Do similar things happen in different materials?
The mechanism observed by the scientists in Stuttgart is reversible, Maier emphasizes. It does not destroy the material. Only when the perovskite material is in contact with a substance that bonds iodine permanently, or when iodine escapes into the atmosphere, does the material degrade over time?
In the near future, the researchers do not have the intention to be content with only understanding the mechanisms of degradation and ultimately preventing them. What is more important, according to Joachim Maier, is to investigate the effect itself, because it represents a novelty in solid-state research. “We will examine other materials to see if similar phenomena occur,” says Joachim Maier. The researchers are also focussing on the question of how this effect can be exploited technically. To do this, they will first develop ideas, such as utilizing light-stimulated storage, and then look for suitable materials for such applications. “Ionic conductivity represents a key phenomenon in an energy research context,” says Joachim Maier. “But in many respects – especially when it comes to exposure to light – it remains terra incognita.” The Max Planck researchers in Stuttgart intend to change this.
Reference: Gee Yeong Kim, et al., “Large tunable photoeffect on ion conduction in halide perovskites and implications for photodecomposition” by Gee Yeong Kim, Alessandro Senocrate, Tae-Youl Yang, Giuliano Gregori, Michael Grätzel and Joachim Maier, 19 March 2018, Nature Materials.
DOI: 10.1038/s41563-018-0038-0
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.