
Sodium (Na), being more than 500 times as abundant as lithium (Li), has recently attracted considerable interest for its potential use in sodium-ion battery technologies.
However, existing sodium-ion batteries face fundamental limitations, including lower power output, constrained storage properties, and longer charging times, necessitating the development of next-generation energy storage materials.
On the 11th of April, KAIST (represented by President Kwang Hyung Lee) announced that a research team led by Professor Jeung Ku Kang from the Department of Materials Science and Engineering had developed a high-energy, high-power hybrid sodium-ion battery capable of rapid charging.
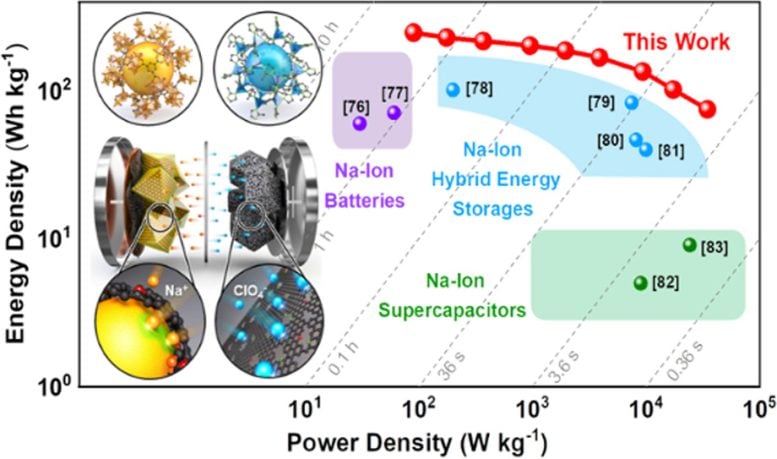
The innovative hybrid energy storage system integrates anode materials typically used in batteries with cathodes suitable for supercapacitors. This combination allows the device to achieve both high storage capacities and rapid charge-discharge rates, positioning it as a viable next-generation alternative to lithium-ion batteries.
Addressing Technological Challenges
However, the development of a hybrid battery with high energy and high power density requires an improvement to the slow energy storage rate of battery-type anodes as well as the enhancement of the relatively low capacity of supercapacitor-type cathode materials.
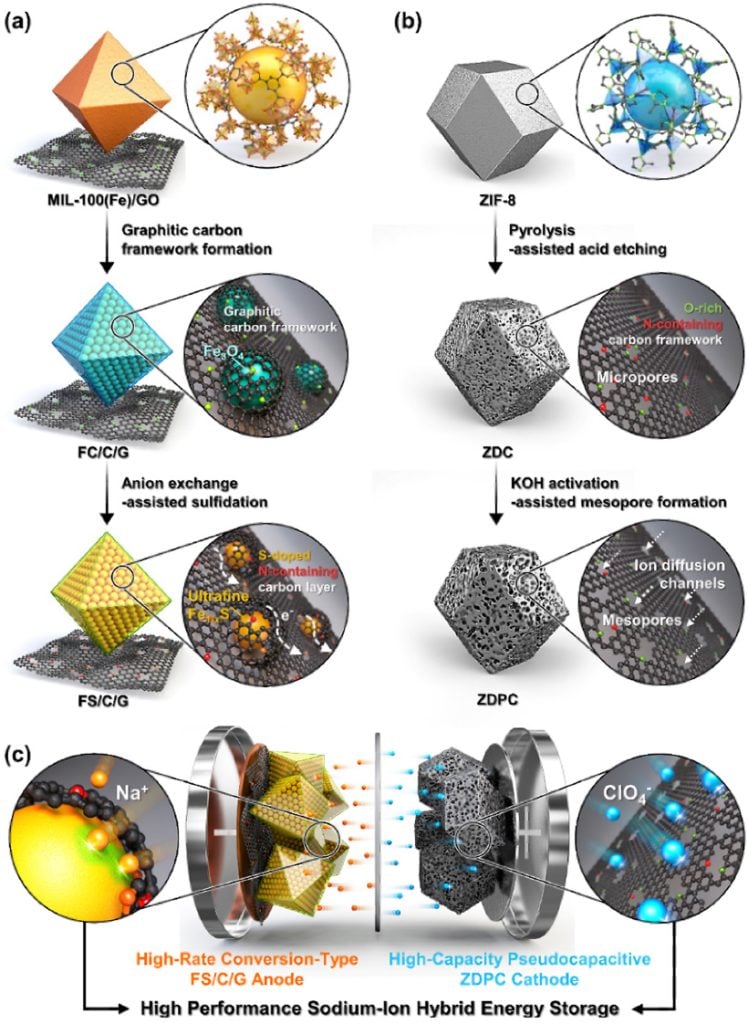
To account for this, Professor Kang’s team utilized two distinct metal-organic frameworks for the optimized synthesis of hybrid batteries. This approach led to the development of an anode material with improved kinetics through the inclusion of fine active materials in porous carbon derived from metal-organic frameworks.
Additionally, a high-capacity cathode material was synthesized, and the combination of the cathode and anode materials allowed for the development of a sodium-ion storage system optimizing the balance and minimizing the disparities in energy storage rates between the electrodes.
Performance and Potential Applications
The assembled full cell, comprising the newly developed anode and cathode, forms a high-performance hybrid sodium-ion energy storage device. This device surpasses the energy density of commercial lithium-ion batteries and exhibits the characteristics of supercapacitors’ power density. It is expected to be suitable for rapid charging applications ranging from electric vehicles to smart electronic devices and aerospace technologies.
Professor Kang noted that the hybrid sodium-ion energy storage device, capable of rapid charging and achieving an energy density of 247 Wh/kg and a power density of 34,748 W/kg, represents a breakthrough in overcoming the current limitations of energy storage systems. He anticipates broader applications across various electronic devices, including electric vehicles.
Reference: “Low-crystallinity conductive multivalence iron sulfide-embedded S-doped anode and high-surface area O-doped cathode of 3D porous N-rich graphitic carbon frameworks for high-performance sodium-ion hybrid energy storages” by Jong Hui Choi, Dong Won Kim, Do Hwan Jung, Keon-Han Kim, Jihoon Kim and Jeung Ku Kang, 27 March 2024, Energy Storage Materials.
DOI: 10.1016/j.ensm.2024.103368
The study was conducted with support from the Ministry of Science and ICT and the National Research Foundation of Korea through the Nanomaterial Technology Development Project.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
3 Comments
Hey SciTech!
How about quantifying things a bit for us poor mortals? It seems articles continually claim something without putting it into perspective. For instance, this article claims,
“hybrid sodium-ion energy storage device, capable of rapid charging and achieving an energy density of 247 Wh/kg and a power density of 34,748 W/kg”
It would be nice to know what reg’lar lithium-ion batteries’ specs are…
Whenever I hear something like this or that technology will allow batteries to charge in seconds, I have to do the math. So, a 40KWh (Kilowatt hour)battery in a Tesla S3 takes 40KWh to completely charge. Lets say that this magic technology can recharge in 5 seconds. So that is 3600(1hr)/5=720. So the charging rate will be 720 * 40KWh = 28,800,000 Watts for 5 seconds. Yeah, that’s almost 29 MEGA WATTS, Yeikes! That’s the power consumed by a small city for 5 seconds. That is 120,000 Amps at 240 Volts. This kind of current would turn any charging cable into plasma in milliseconds, NOT GOOD! So this claim is patently impossible by the laws of physics. When we discuss the ability to recharge at a fast rate, we need to keep reality in mind.
W I am never gonna try this bro oh yo, ho, doll toy toy
Never use this it’s really bad just be careful careful but I saw it on YouTube, but I’m only six years old so bro, I don’t know what to do what to say actually bro I mean, bro