
The CRISPR-based gene editor, C-to-G Base Editor (CGBE), opens up treatment avenues for up to 40 percent of genetic disorders caused by single-nucleotide mutations.
A team of researchers from the Agency for Science, Technology and Research’s (A*STAR) Genome Institute of Singapore (GIS) has developed a CRISPR-based gene editor, C-to-G Base Editor (CGBE), to correct mutations that cause genetic disorders. Their research was published on March 2, 2021, in the journal Nature Communications.
One in seventeen people in the world suffers from some type of genetic disorder. Chances are, you or someone you know — a relative, friend, or colleague — is one of approximately 450 million people affected worldwide. Mutations responsible for these disorders can be caused by multiple mutagens — from sunlight to spontaneous errors in your cells. The most common mutation by far is the single-based substitution, in which a single-base in the DNA (such as G) is replaced by another base (such as C). Countless cystic fibrosis patients worldwide have C instead of G, leading to defective proteins that cause the genetic disease. In another case, replacing A with T in hemoglobin causes sickle cell anemia.
To fix these substitutions, the team invented a CRISPR-based gene editor that precisely changes the defective C within the genome to the desired G. This C-to-G base editor (CGBE) invention opens up treatment options for approximately 40 percent of the single-base substitutions that are associated with human diseases such as the aforementioned cystic fibrosis, cardiovascular diseases, musculoskeletal diseases, and neurological disorders.
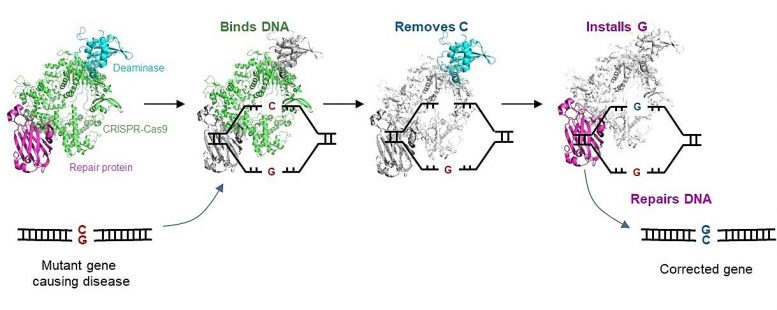
The CGBE editor advances the widely adopted CRISPR-Cas9 technology to enable molecular surgery on the human genome. The CRISPR-Cas9 technology is routinely used to disrupt target genes, but it is inefficient when a precise change to particular sequences is desired. The CGBE editor resolves a key aspect of this challenge by enabling efficient and precise genetic changes. CGBE consists of three parts: 1) a modified CRISPR-Cas9 will pinpoint the mutant gene and focus the entire editor on that gene; 2) a deaminase (an enzyme that removes the amino group from a compound) will then target the defective C, and mark it for replacement, and 3) finally, a protein will initiate cellular mechanisms to replace that defective C with a G. This enables a previously unachievable direct conversion from C to G, correcting the mutation and, consequently, treating the genetic disorder.
Dr. Chew Wei Leong, Senior Research Scientist at GIS, said, “The CGBE gene editor is a ground-breaking invention that for the first time, directly converts C to G in genes, which potentially opens up treatment avenues for a substantial fraction of genetic disorders associated with single-nucleotide mutations.”
“The safety of patients is critical,” Dr. Chew emphasized. “We are working to ensure our CGBE and CRISPR-Cas modalities are both effective and safe in disease models before we can further develop such modalities for the clinic.” For his scientific endeavors in gene editing therapy, he was one of the three young researchers who clinched the prestigious Young Scientist Award (YSA) 2020.
Prof Patrick Tan, Executive Director of GIS, said, “Novel editors such as CGBE are expanding the growing suite of precise genome-editing tools that include cytidine base editors (CBEs), adenine base editors (ABEs), CGBEs, and prime editors. Together, they enable the precise and efficient engineering of DNA for research, biological interrogation, and disease correction, thereby ushering a new age of genetic medicine.”
Reference: “Programmable C:G to G:C genome editing with CRISPR-Cas9-directed base excision repair proteins” by Liwei Chen, Jung Eun Park, Peter Paa, Priscilla D. Rajakumar, Hong-Ting Prekop, Yi Ting Chew, Swathi N. Manivannan and Wei Leong Chew, 2 March 2021, Nature Communications.
DOI: 10.1038/s41467-021-21559-9
About A*STAR’s Genome Institute of Singapore (GIS)
The Genome Institute of Singapore (GIS) is an institute of the Agency for Science, Technology and Research (A*STAR). It has a global vision that seeks to use genomic sciences to achieve extraordinary improvements in human health and public prosperity. Established in 2000 as a center for genomic discovery, the GIS pursues the integration of technology, genetics and biology towards academic, economic, and societal impact, with a mission to “read, reveal and write DNA for a better Singapore and world.”
Key research areas at the GIS include Precision Medicine & Population Genomics, Genome Informatics, Spatial & Single Cell Systems, Epigenetic & Epitranscriptomic Regulation, Genome Architecture & Design, and Sequencing Platforms. The genomics infrastructure at the GIS is also utilized to train new scientific talent, to function as a bridge for academic and industrial research, and to explore scientific questions of high impact.
About the Agency for Science, Technology and Research (A*STAR)
The Agency for Science, Technology and Research (A*STAR) is Singapore’s lead public sector agency that spearheads economic-oriented research to advance scientific discovery and develop innovative technology. Through open innovation, we collaborate with our partners in both the public and private sectors to benefit society.
As a Science and Technology Organisation, A*STAR bridges the gap between academia and industry. Our research creates economic growth and jobs for Singapore, and enhances lives by contributing to societal benefits such as improving outcomes in healthcare, urban living, and sustainability.
We play a key role in nurturing and developing a diversity of talent and leaders in our Agency and research entities, the wider research community and industry. A*STAR’s R&D activities span biomedical sciences and physical sciences and engineering, with research entities primarily located in Biopolis and Fusionopolis.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
4 Comments
Would this help people with CF? Or embryos with CF? ie would it edit every cell in a human’s body?
One thing I don’t understand: How is a single edited gene going to provide relief? How is it copied into all the relevant cells?
Reply to Rebeka Love: Good point, one edited cell wouldn’t be enough. One must be innovative. 1. Deploy a vector that delivers fix to many. 2. Extract stem cells, fix and culture in vitro then put back and let them reproduce and produce desired tissue. 3. Fix a gamete that becomes a zygote or embryonic cell which divides copying the fix into many.
Could this be used for epilepsy in any way?