Scientists have uncovered how aging alters tryptophan metabolism in the brain, linking a longevity protein to neurodegeneration.
Tryptophan is often associated with sleep, but its role in the body extends far beyond that reputation. This essential amino acid serves as a foundational building block for proteins, supports cellular energy production through (NAD+), and provides the raw material needed to produce important brain chemicals such as serotonin and melatonin. Together, these pathways help regulate mood, cognition, and normal sleep cycles.
As the brain ages or becomes affected by neurological disease, this finely tuned system begins to break down. Studies have shown that tryptophan catabolism becomes increasingly dysregulated in aging brains and even more so in neurodegenerative and psychiatric conditions.
These metabolic shifts are linked to worsening mood regulation, impaired learning, and disrupted sleep behavior. Although the consequences of this imbalance have been well documented, the underlying molecular processes that drive changes in how tryptophan is used remained unclear.
Identifying the Molecular Trigger
Prof. Debra Toiber and her colleagues at Ben-Gurion University of the Negev have uncovered a specific biological explanation for this disruption. Their work points to the absence of a longevity-linked protein known as Sirtuin 6 (SIRT6) as a central factor in the loss of normal metabolic control.
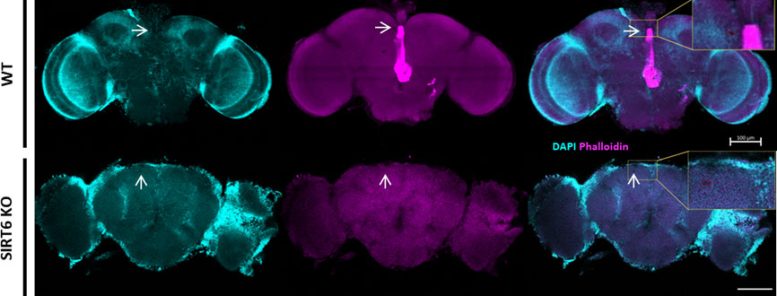
Using experiments in cells, Drosophila (fly), and mice, the researchers showed that SIRT6 plays an active role in directing gene activity, including genes such as (e.g., TDO2, AANAT). When SIRT6 is missing, this genetic regulation shifts tryptophan metabolism toward the kynurenic pathway, which is associated with neurotoxic effects, while reducing the production of neurotransmitters that normally support and protect brain function.

Their findings were just published in Nature Communications.
A Reversible Process With Therapeutic Potential
Crucially, Prof. Toiber and her team show that this damage is not permanent. By inhibiting the enzyme TDO2 in their SIRT6 knockout fly model, they were able to significantly reverse both neuromotor decline and vacuolar formation, indicating a powerful therapeutic window.
“Our research positions SIRT6 as a critical, upstream drug target for combating neurodegenerative pathology,” says Prof. Toiber.
Reference: “Histone deacetylase SIRT6 regulates tryptophan catabolism and prevents metabolite imbalance associated with neurodegeneration” by Shai Kaluski-Kopatch, Daniel Stein, Alfredo Garcia Venzor, Ana Margarida Ferreira Campos, Melanie Planque, Bareket Goldstein, Estefanía De Allende-Becerra, Dmitrii Smirnov, Adam Zaretsky, Ekaterina Eremenko, Miguel Portillo, Monica Einav, Alena Bruce Krejci, Uri Abdu, Ekaterina Khrameeva, Daniel Gitler, Sarah-Maria Fendt and Debra Toiber, 4 December 2025, Nature Communications.
DOI: 10.1038/s41467-025-67021-y
The study was supported by the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation program (grant agreement No 849029), the David and Inez Myers foundation, the Israeli Ministry of Science and Technology (MOST), the High-tech, Bio-tech and Negev fellowships of Kreitman School of Advanced Research of Ben-Gurion University and The Israel Science Foundation (Grant no. 422/23). The RNA-seq data analysis was supported by the Russian Science Foundation (grant number 25-71-20017).
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.