Identification of a Molecular Code Embedded in Protein for Regulating Its Glycosylation
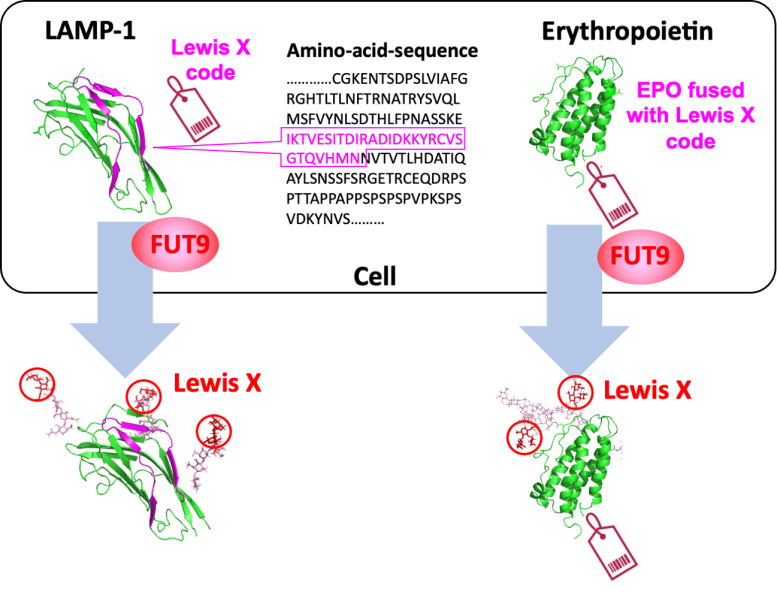
Many proteins in nature exist as glycoproteins, which are molecules comprised of protein (polypeptide chain) and glycan (sugar chain). While the protein structure is determined on the basis of its genetic blueprint, the information on glycans is not directly encoded by the genome. Therefore, it is quite challenging to control protein glycosylation. Under these circumstances, a collaborative research group, including scientists at Nagoya City University, National Institutes of Natural Sciences, and Academia Sinica, has found a specific amino acid sequence in a polypeptide that induces a specific glycan structure called Lewis X.
The group of scientists in Japan has previously shown that Lewis X specifically modifies the protein LAMP-1 in mouse neural stem cells through the enzymatic action of fucosyltransferase 9 (FUT9). In this recent research, they have shown that Lewis X modification specific for LAMP-1 occurs not only in neural stem cells but also in several cultured mammalian cells. Furthermore, they also discovered that a sequence consisting of 29 amino-acid residues in LAMP-1 promotes Lewis X modification catalyzed by the enzyme, and this sequence induces Lewis X modification when fused to other proteins used as biopharmaceuticals. This means that, in glycoprotein molecules, a specific amino-acid sequence can determine their glycan structures.
Most biopharmaceuticals are actually glycoproteins, as best exemplified by therapeutic antibodies. Their glycan structures are critical for their efficacy and safety. Therefore, the control of glycosylation is an important issue in the development of biopharmaceuticals. Their findings on a regulatory code of protein glycosylation are expected to pave the way for controlling glycosylation of biopharmaceuticals.
Reference: “An embeddable molecular code for Lewis X modification through interaction with fucosyltransferase 9” by Taiki Saito, Hirokazu Yagi, Chu-Wei Kuo, Kay-Hooi Khoo and Koichi Kato, 13 July 2022, Communications Biology.
DOI: 10.1038/s42003-022-03616-1
Funding: Japan Science and Technology Agency, Grants-in-Aid for Scientific Research
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.