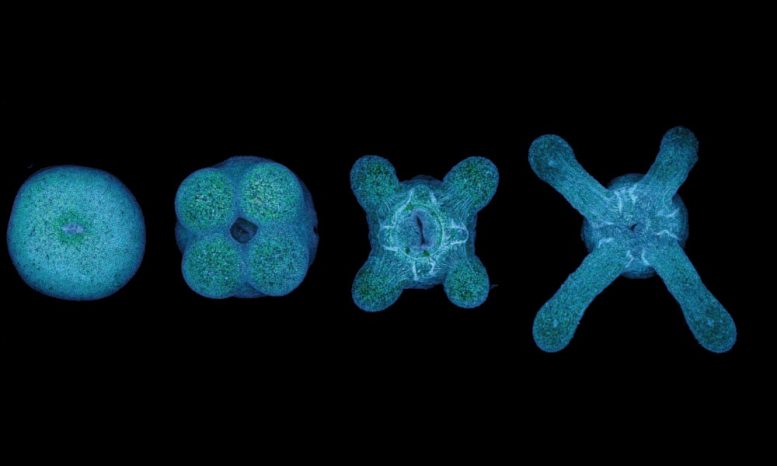
Your genetic code determines that you will grow two arms and two legs. The same fate is true for all mammals. Similarly, the number of fins a fish has and the number of legs and wings an insect has are embedded in their genetic code. Sea anemones, however, defy this rule and have a variable number of tentacle arms.
Until now it’s been unclear what regulates the number of tentacles a sea anemone can grow. Scientists from the Ikmi group at EMBL Heidelberg, in collaboration with researchers in the Gibson lab at the Stowers Institute for Medical Research in Kansas City, have shown that the number of tentacles is defined by the amount of food consumed. “Controlling the number of tentacle arms by food intake makes the sea anemone behave more like a plant developing new branches than an animal growing a new limb,” explains group leader Aissam Ikmi. Defining what environmental factors trigger morphological changes is a particularly important question given the longevity of sea anemones, with some species living for more than 65 years. “As predominantly sessile animals, sea anemones must have evolved strategies to deal with environmental changes to sustain such a long lifespan,” adds Ikmi.
The scientists have shown that the growth of new tentacles happens not only when the sea anemone is a juvenile, but also throughout adulthood. “We can conclude that the number of tentacle arms must be determined by the interplay between genetic and environmental factors,” says Ikmi, who started this project when he was still a postdoc in the lab of Matt Gibson. While the sea anemone uses different strategies to build tentacles in the different stages of its life, the final arms are morphologically indistinguishable from each other. “If humans could do the same, it would mean that the more we ate, the more arms and legs we could grow,” says Ikmi. “Imagine how handy it would be if we could activate this when we needed to replace damaged limbs.”
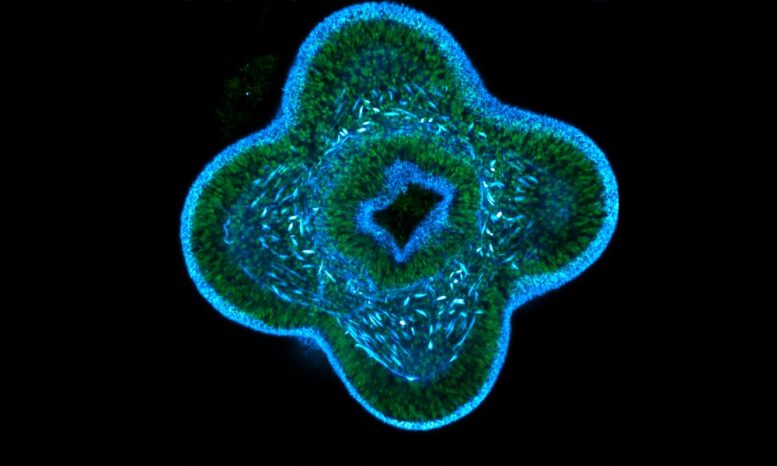
When Ikmi’s group studied the locations at which the new arms form, they found that muscle cells pre-mark the sites of new tentacles. These muscle cells change their gene expression signature in response to food. The same molecular signaling employed to build tentacles in sea anemones also exists in many other species — including humans. So far, however, its role has been studied mainly in embryonic development. “We propose a new biological context in which to understand how nutrient uptake impacts the function of this developmental signaling: a situation that is relevant for defining the role of metabolism in guiding the formation of organs during adulthood” explains Ikmi. “Sea anemones show us that it is possible that nutrients are not converted into excess fat storage — as it is the case in all mammals — but instead transformed into a new body structure.”
While this finding is novel on its own, it also shows that sea anemones, which are traditionally used for evolutionary developmental studies, are well suited to study morphogenesis in the context of organism-environment interactions.
To build the branching map of new tentacles, researchers analyzed more than 1000 sea anemones one by one. “Scoring such a massive number of tentacles is, in some ways, a story in itself,” says Mason McMullen, laughing. McMullen, a clinical pharmacist at the University of Kansas Health System, spent months imaging sea anemones’ heads to score their tentacle number and location.
Knowing that the number of tentacles in sea anemones is determined by their food intake, the group plans to define the key nutrients critical to this process. Ikmi and his group also want to further investigate the unconventional role of muscles in defining the sites where new tentacles form. “We’re currently investigating this novel property of muscle cells and are eager to find out the mystery behind them,” he concludes.
Reference: “Feeding-dependent tentacle development in the sea anemone Nematostella vectensis” by Aissam Ikmi, Petrus J. Steenbergen, Marie Anzo, Mason R. McMullen, Anniek Stokkermans, Lacey R. Ellington and Matthew C. Gibson, 2 September 2020, Nature Communications.
DOI: 10.1038/s41467-020-18133-0
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.