A hidden rearrangement of ordinary chlorophyll allows certain algae to access light others cannot.
Life in dim environments pushes photosynthetic organisms to their limits. In places where sunlight is scarce or filtered, some species have evolved ways to use parts of the light spectrum that most others cannot. A new study from Osaka Metropolitan University reveals how a freshwater alga extends its reach into far-red light by reorganizing one of biology’s most familiar molecules.
Far-red light carries less energy than the wavelengths typically used in photosynthesis, which is why most plants and algae rely on visible red and blue light instead. Yet in dense forests or sediment-rich water, those higher-energy wavelengths are often absorbed before they can penetrate deeper layers. What remains is far-red light, and for most organisms, it is not enough.
“Whilst certain cyanobacteria use specialized chlorophylls to absorb far-red light, many plants and algae achieve the same effect by reorganizing ordinary chlorophyll a into cooperative assemblies within their photosynthetic antennas,” said Ritsuko Fujii, lead author and associate professor at the Graduate School of Science and Research Center for Artificial Photosynthesis at Osaka Metropolitan University.
Chlorophyll a is a pigment that cannot absorb far-red light on its own. So, how exactly do these organisms achieve photosynthesis?
A Unique Alga Offers Clues
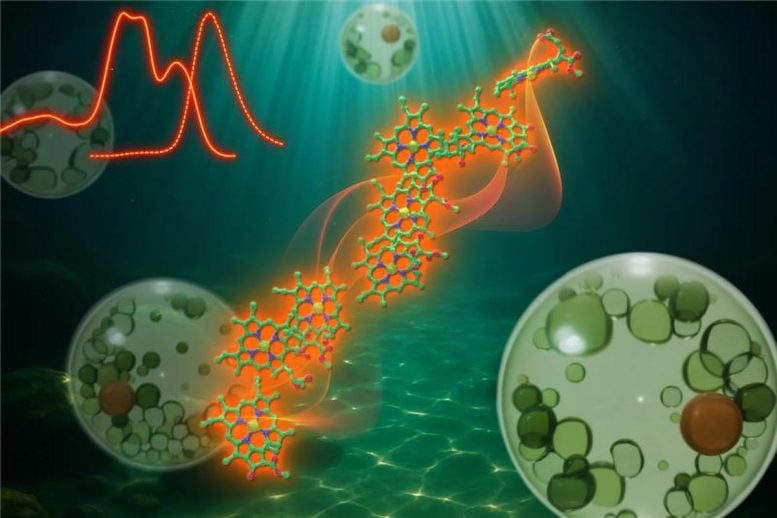
To investigate, the researchers studied the freshwater eustigmatophyte alga Trachydiscus minutus. This species builds up large amounts of a light-harvesting protein that can use far-red light.
While the alga can photosynthesize under normal lighting, these elevated protein levels become especially important in low-light environments.
“The organism produces a specialized photosynthetic antenna called a red-shifted violaxanthin–chlorophyll protein (rVCP), which absorbs far-red light even though it contains only chlorophyll a,” Fujii said.
Using cryo-electron microscopy, the researchers determined the structure of rVCP at a high resolution of 2.4 Å. They found that the protein forms a previously unreported architecture: a tetramer composed of two different heterodimers. This unique assembly brings chlorophyll a molecules into close proximity, allowing them to form unusually large pigment clusters.
To see how this structure influences light absorption, the researchers combined structural data with multiscale quantum chemical calculations.
Energy Sharing Unlocks Far-Red Light
“Our analysis showed that three chlorophyll clusters within each heterodimer play a major role in absorbing far-red light,” Fujii said. “Importantly, this absorption arises purely from energy delocalization across multiple chlorophyll molecules, independently of the charge-transfer effects that are thought to drive similar red-shifted systems.”
The results point to a different way of tuning light absorption. Instead of altering the pigment itself, the protein framework controls how identical chlorophyll molecules interact, allowing them to capture longer wavelengths of light.
This mechanism helps explain how certain organisms continue to function in low-light environments.
Implications for Energy and Design
The findings could also have practical uses. Some eustigmatophytes can store large amounts of oil, making them attractive for sustainable bioenergy production. Organisms that efficiently use far-red light could enable oil production in environments that are currently unsuitable.
The unusual tetramer structure of rVCP may also serve as a model for designing new proteins. Because pigment arrangement depends on protein sequence, this system could help guide efforts to engineer artificial or improved photosynthetic technologies.
“As interest grows in expanding photosynthesis into the far-red region to boost overall photosynthetic productivity on Earth, our next goal is to reveal how this complex delivers captures energy to the photosystem and how that mechanism could be optimized,” Fujii said.
Reference: “Exciton Delocalization Promotes Far-Red Absorption in a Tetrameric Chlorophyll a Light-Harvesting Complex from Trachydiscus minutus” by Soichiro Seki, Lorenzo Cupellini, David Bína, Elena Betti, Petra Urajová, Hideaki Tanaka, Tomoko Miyata, Keiichi Namba, Genji Kurisu, Tomáš Polívka, Radek Litvín and Ritsuko Fujii, 13 December 2025, Journal of the American Chemical Society.
DOI: 10.1021/jacs.5c17299
Funding: Japan Society for the Promotion of Science, Japan Society for the Promotion of Science, Japan Science and Technology Agency, Japan Agency for Medical Research and Development, Osaka University, Institutional support RVO
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.